GPI锚定丝氨酸蛋白酶testisin的活化机制研究
Mechanistic studies on regulation of the activity of GPI-anchored serine protease testisin
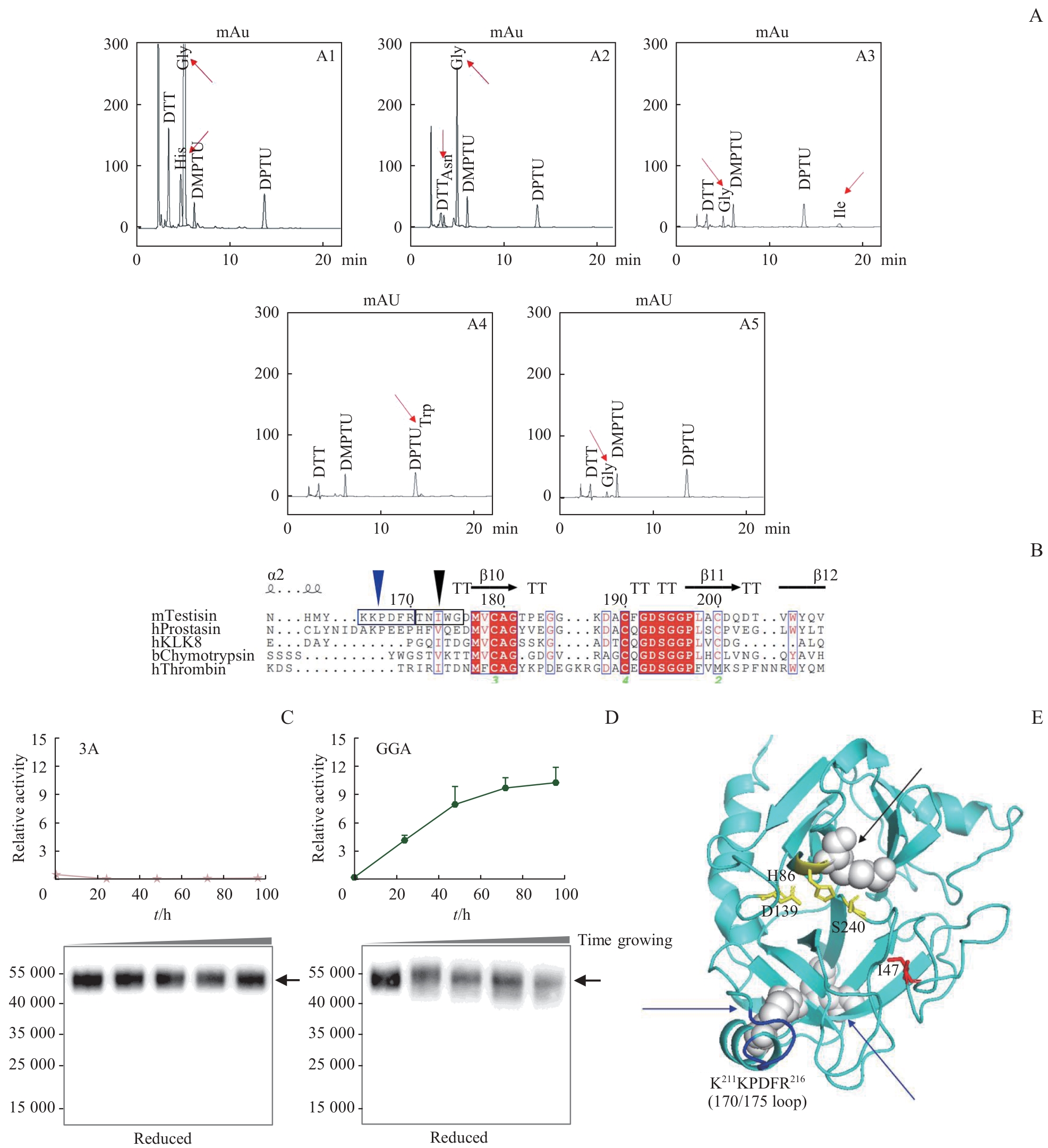
Note:A. The results for the N-terminal sequence of the light chain peptide of mTN (the first five amino acids). The red arrow indicates the measured amino acid signals. The Y-axis represents signal intensity, and the X-axis represents the testing time. Au—Absorbance unit.1 Au=1 000 mAu. B. The comparison result of the catalytic domain sequences of mTN with other serine proteases (partial). The five amino acids indicated by the black triangle corresponded to the first five amino acids of the N-terminal sequencing; the six amino acids indicated by the blue triangle corresponded to amino acids 211?216 (KKPDFR) of the recombinant protein, where the cleavage site was thought to be located. C.The enzymatic activity trend and Western blotting result of the 3A mutant. D. The enzymatic activity trend and Western blotting result of the GGA mutant. E. Predicted structure of the mTN zymogen generated by Alphafold 2.