
上海交通大学学报(医学版) ›› 2021, Vol. 41 ›› Issue (12): 1603-1611.doi: 10.3969/j.issn.1674-8115.2021.12.009
沈剑箫1( ), 王万鹏2, 邵兴华1, 吴晶魁1, 李舒1, 车霞静1, 倪兆慧1(
), 王万鹏2, 邵兴华1, 吴晶魁1, 李舒1, 车霞静1, 倪兆慧1( )
)
收稿日期:2021-08-12
出版日期:2021-12-28
发布日期:2021-01-28
通讯作者:
倪兆慧,电子信箱:profnizh@126.com。作者简介:沈剑箫(1985—),男,主治医师,博士;电子信箱:shenjianxiao@aliyun.com。
基金资助:
Jian-xiao SHEN1( ), Wan-peng WANG2, Xing-hua SHAO1, Jing-kui WU1, Shu LI1, Xia-jing CHE1, Zhao-hui NI1(
), Wan-peng WANG2, Xing-hua SHAO1, Jing-kui WU1, Shu LI1, Xia-jing CHE1, Zhao-hui NI1( )
)
Received:2021-08-12
Online:2021-12-28
Published:2021-01-28
Contact:
NI Zhao-hui, E-mail: profnizh@126.com.Supported by:摘要:
目的·探讨N6-甲基腺嘌呤(N6-methyladenosine,m6A)甲基化修饰在顺铂诱导的小鼠急性肾损伤进程中的作用。方法·选择4只C57bL/6小鼠,尾静脉注射顺铂(20 mg/kg)诱导急性肾损伤(损伤组);另取4只C57bL/6小鼠,尾静脉注射等量生理盐水(对照组)。检测2组小鼠血清肌酐及血尿素氮水平变化,观察小鼠肾脏组织切片中病理损伤情况,评估模型是否成功。进一步运用甲基化RNA免疫共沉淀技术(methylated RNA immunoprecipitation,MeRIP)与RNA测序技术分别检测2组小鼠肾脏组织中m6A甲基化水平与RNA表达变化。运用基因本体论及京都基因和基因组数据库进行结果可视化和综合研究,并将RNA测序技术所得转录组数据与MeRIP技术检测所得表观遗传数据联合分析,寻找参与顺铂诱导急性肾损伤病理变化过程的候选基因。结果·顺铂可诱导小鼠血清肌酐与血尿素氮水平显著升高。光学显微镜观察肾组织发现广泛的肾小管空泡变性,上皮细胞剥脱,肾小管坏死,提示造模成功。MeRIP检测发现损伤组与对照组小鼠肾脏中共有2 227个基因含有2 981个差异化表达的m6A甲基化位点(表达变化倍数≥2且P<0.05),这些基因主要富集于代谢及细胞死亡通路。表达差异化m6A甲基化位点的基因与RNA差异化表达基因的联合分析发现1 002个表达趋势相同的基因,如纤维蛋白原α链、溶质载体12家族成员1和甲肝病毒细胞受体1等。结论·顺铂可诱导肾脏组织中基因mRNA上m6A甲基化位点的甲基化水平变化,促进急性肾损伤进程。
中图分类号:
沈剑箫, 王万鹏, 邵兴华, 吴晶魁, 李舒, 车霞静, 倪兆慧. 顺铂诱导的急性肾损伤中肾脏组织m6A甲基化水平的变化[J]. 上海交通大学学报(医学版), 2021, 41(12): 1603-1611.
Jian-xiao SHEN, Wan-peng WANG, Xing-hua SHAO, Jing-kui WU, Shu LI, Xia-jing CHE, Zhao-hui NI. Changes of m6A methylation in renal tissue during cisplatin-induced acute injury[J]. JOURNAL OF SHANGHAI JIAOTONG UNIVERSITY (MEDICAL SCIENCE), 2021, 41(12): 1603-1611.

图1 顺铂诱导的C57bL/6小鼠AKI模型建立Note: A. Analysis of serum creatinine level in mice following different treatments.①P=0.000. B. Analysis of urea nitrogen level in mice following different treatments. ②P=0.000. C. Image of hematoxylin and eosin staining in kidney (black arrows indicating the injury). D. Score for characteristic histologic signs of renal injury. ③P=0.000.
Fig 1 Establishment of CI-AKI model in C57bL/6 mice
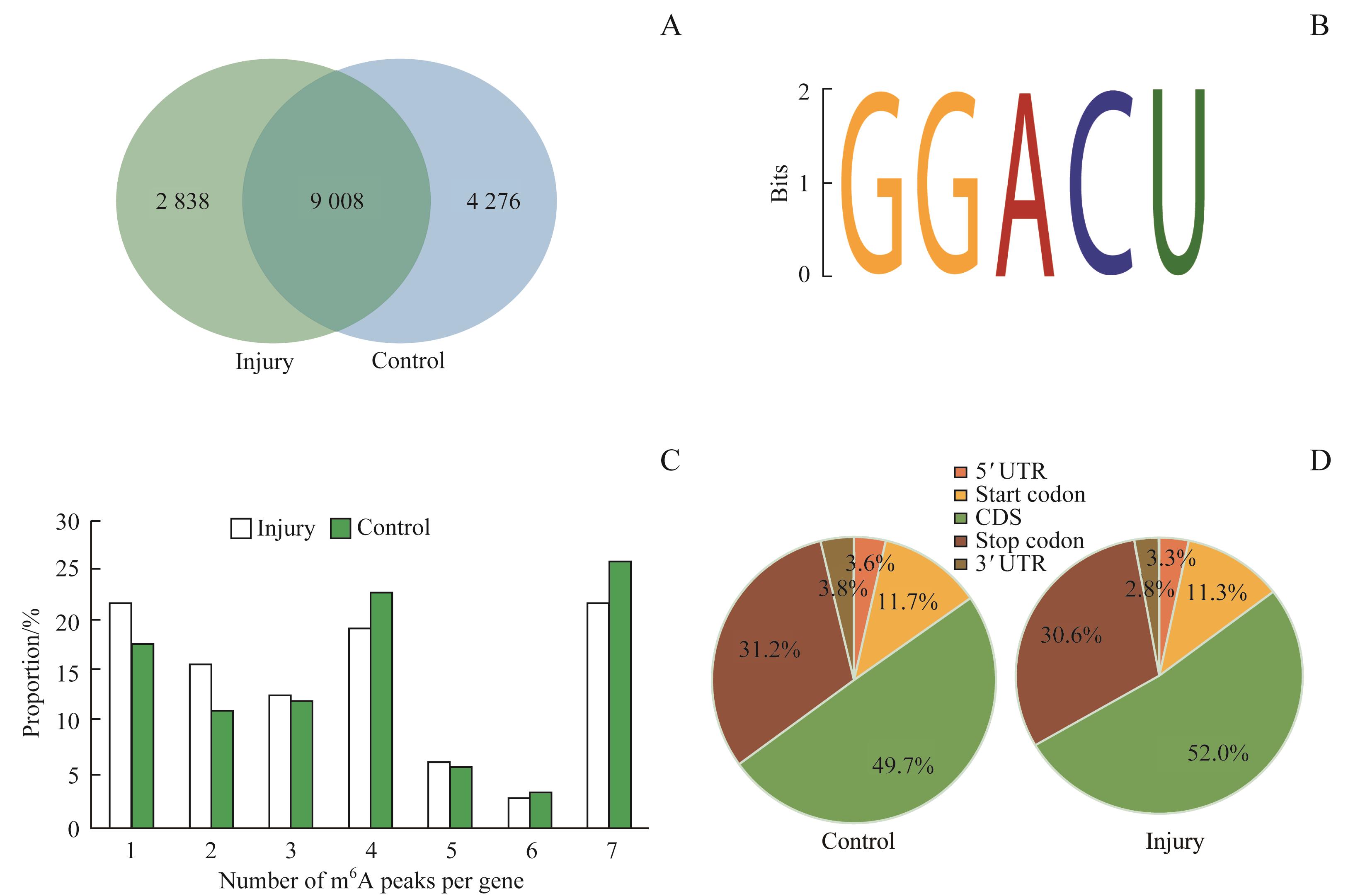
图2 对照组和损伤组mRNAs中m6A变化情况Note: A. Venn diagram showing the overlap of m6A peaks within mRNAs between the injury group and the control group. B. The top motif enriched across m6A peaks was identified from both groups. C. Proportion of genes harboring different numbers of m6A peaks in both groups. D. Pie charts showing the percentage of m6A peaks in five segments of transcripts. m6A peaks were most enriched in the coding sequence segment.
Fig 2 Overview of m6A methylation within mRNAs in the control group and the injury group
| Gene name | Full name | Gene ID | Fold change | Regulation | Chromsome | Peak start | Peak end | Peak length | P value |
|---|---|---|---|---|---|---|---|---|---|
| BC061237 | cDNA sequence BC061237 | 385138 | 1 489.5 | Up | Chr14 | 44504106 | 44504291 | 185 | 0.000 |
| BC061237 | cDNA sequence BC061237 | 385138 | 816.8 | Up | Chr14 | 44500121 | 44500197 | 76 | 0.000 |
| Krt20 | Keratin 20 | 66809 | 498.7 | Up | Chr11 | 99430752 | 99430920 | 168 | 0.000 |
| Krt20 | Keratin 20 | 66809 | 498.0 | Up | Chr11 | 99429021 | 99429078 | 57 | 0.000 |
| Krt20 | Keratin 20 | 66809 | 483.5 | Up | Chr11 | 99432181 | 99432343 | 162 | 0.000 |
| 1700001F09Rik | RIKEN cDNA 1700001F09 gene | 71826 | 383.8 | Up | Chr14 | 43346701 | 43346790 | 89 | 0.000 |
| Ccdc85b | Coiled-coil domain containing 85B | 240514 | 328.6 | Up | Chr19 | 5454141 | 5454580 | 439 | 0.000 |
| Gm3543 | Predicted gene 3543 | 100041849 | 318.0 | Up | Chr14 | 41982201 | 41982290 | 89 | 0.000 |
| Serpina3n | Serinepeptidase inhibitor, clade A, member 3N | 20716 | 316.3 | Up | Chr12 | 104414261 | 104414329 | 68 | 0.000 |
| Gm3543 | Predicted gene 3543 | 100041849 | 254.0 | Up | Chr14 | 41982133 | 41982180 | 47 | 0.000 |
| Alms1 | ALMS1 centrosome and basal body associated protein | 236266 | 503.6 | Down | Chr6 | 85694833 | 85694951 | 118 | 0.000 |
| Tas2r119 | Taste Receptor, type 2, member 119 | 57254 | 385.6 | Down | Chr15 | 32177288 | 32177620 | 332 | 0.000 |
| Ctnna2 | Catenin α 2 | 12386 | 220.8 | Down | Chr6 | 77600041 | 77600380 | 339 | 0.000 |
| Afm | Afamin | 280662 | 195.1 | Down | Chr5 | 90518931 | 90519060 | 129 | 0.000 |
| Slc5a4a | Solute carrier family 5, member 4a | 64452 | 126.7 | Down | Chr10 | 76163688 | 76163769 | 81 | 0.000 |
| Nat1 | N-acetyltransferase 1 | 17960 | 123.5 | Down | Chr8 | 67490861 | 67491460 | 599 | 0.000 |
| Pzp | PZP α-2-macroglobulin like | 11287 | 111.1 | Down | Chr6 | 128526621 | 128526720 | 99 | 0.000 |
| Dpf3 | Double PHD fingers 3 | 70127 | 99.2 | Down | Chr12 | 83215461 | 83215800 | 339 | 0.000 |
| Dgkg | Diacylglycerol Kinase γ | 110197 | 95.3 | Down | Chr16 | 22479365 | 22479426 | 61 | 0.000 |
| Dpf3 | Double Phd fingers 3 | 70127 | 94.3 | Down | Chr12 | 83214541 | 83214840 | 299 | 0.000 |
表1 甲基化水平变化差异最大的20个m6A甲基化位点
Tab 1 Top 20 differentially methylated m6A peaks
| Gene name | Full name | Gene ID | Fold change | Regulation | Chromsome | Peak start | Peak end | Peak length | P value |
|---|---|---|---|---|---|---|---|---|---|
| BC061237 | cDNA sequence BC061237 | 385138 | 1 489.5 | Up | Chr14 | 44504106 | 44504291 | 185 | 0.000 |
| BC061237 | cDNA sequence BC061237 | 385138 | 816.8 | Up | Chr14 | 44500121 | 44500197 | 76 | 0.000 |
| Krt20 | Keratin 20 | 66809 | 498.7 | Up | Chr11 | 99430752 | 99430920 | 168 | 0.000 |
| Krt20 | Keratin 20 | 66809 | 498.0 | Up | Chr11 | 99429021 | 99429078 | 57 | 0.000 |
| Krt20 | Keratin 20 | 66809 | 483.5 | Up | Chr11 | 99432181 | 99432343 | 162 | 0.000 |
| 1700001F09Rik | RIKEN cDNA 1700001F09 gene | 71826 | 383.8 | Up | Chr14 | 43346701 | 43346790 | 89 | 0.000 |
| Ccdc85b | Coiled-coil domain containing 85B | 240514 | 328.6 | Up | Chr19 | 5454141 | 5454580 | 439 | 0.000 |
| Gm3543 | Predicted gene 3543 | 100041849 | 318.0 | Up | Chr14 | 41982201 | 41982290 | 89 | 0.000 |
| Serpina3n | Serinepeptidase inhibitor, clade A, member 3N | 20716 | 316.3 | Up | Chr12 | 104414261 | 104414329 | 68 | 0.000 |
| Gm3543 | Predicted gene 3543 | 100041849 | 254.0 | Up | Chr14 | 41982133 | 41982180 | 47 | 0.000 |
| Alms1 | ALMS1 centrosome and basal body associated protein | 236266 | 503.6 | Down | Chr6 | 85694833 | 85694951 | 118 | 0.000 |
| Tas2r119 | Taste Receptor, type 2, member 119 | 57254 | 385.6 | Down | Chr15 | 32177288 | 32177620 | 332 | 0.000 |
| Ctnna2 | Catenin α 2 | 12386 | 220.8 | Down | Chr6 | 77600041 | 77600380 | 339 | 0.000 |
| Afm | Afamin | 280662 | 195.1 | Down | Chr5 | 90518931 | 90519060 | 129 | 0.000 |
| Slc5a4a | Solute carrier family 5, member 4a | 64452 | 126.7 | Down | Chr10 | 76163688 | 76163769 | 81 | 0.000 |
| Nat1 | N-acetyltransferase 1 | 17960 | 123.5 | Down | Chr8 | 67490861 | 67491460 | 599 | 0.000 |
| Pzp | PZP α-2-macroglobulin like | 11287 | 111.1 | Down | Chr6 | 128526621 | 128526720 | 99 | 0.000 |
| Dpf3 | Double PHD fingers 3 | 70127 | 99.2 | Down | Chr12 | 83215461 | 83215800 | 339 | 0.000 |
| Dgkg | Diacylglycerol Kinase γ | 110197 | 95.3 | Down | Chr16 | 22479365 | 22479426 | 61 | 0.000 |
| Dpf3 | Double Phd fingers 3 | 70127 | 94.3 | Down | Chr12 | 83214541 | 83214840 | 299 | 0.000 |
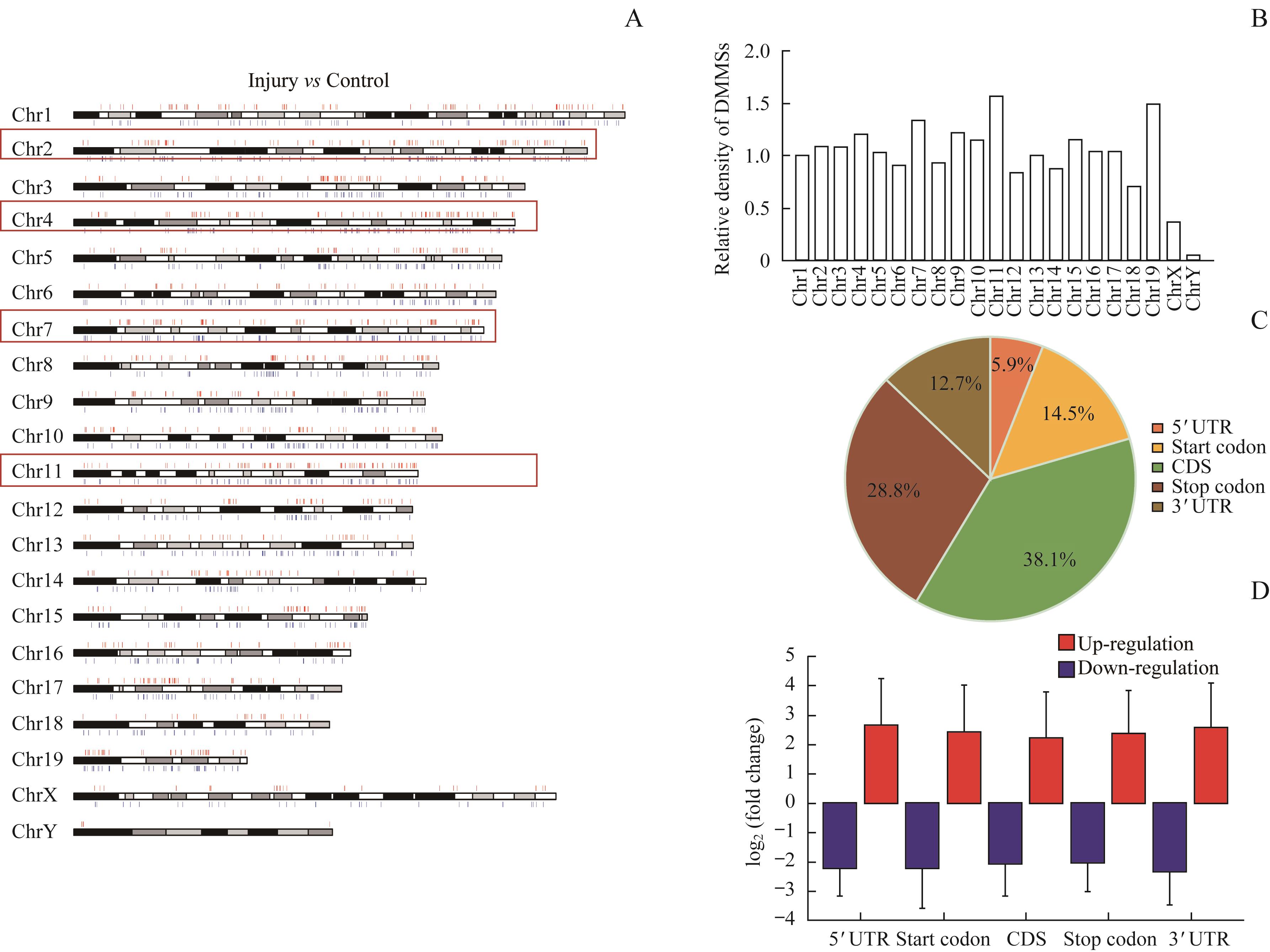
图3 DMMS在染色体上分布情况Note: A. Chromosomal distribution of all DMMSs. Red lines indicate up regulation m6A methylation sites; Blue line indicate down regulation m6A methylation sites. B. Relative occupancy of DMMSs in each chromosome was normalized by length. C. Pie chart showing the percentage of DMMS peaks in five non-overlapping segments. D. Statistics of fold change of DMMS peaks in five segments. The histogram shows the mean of the fold change.
Fig 3 Distribution of DMMSs in chromosomes
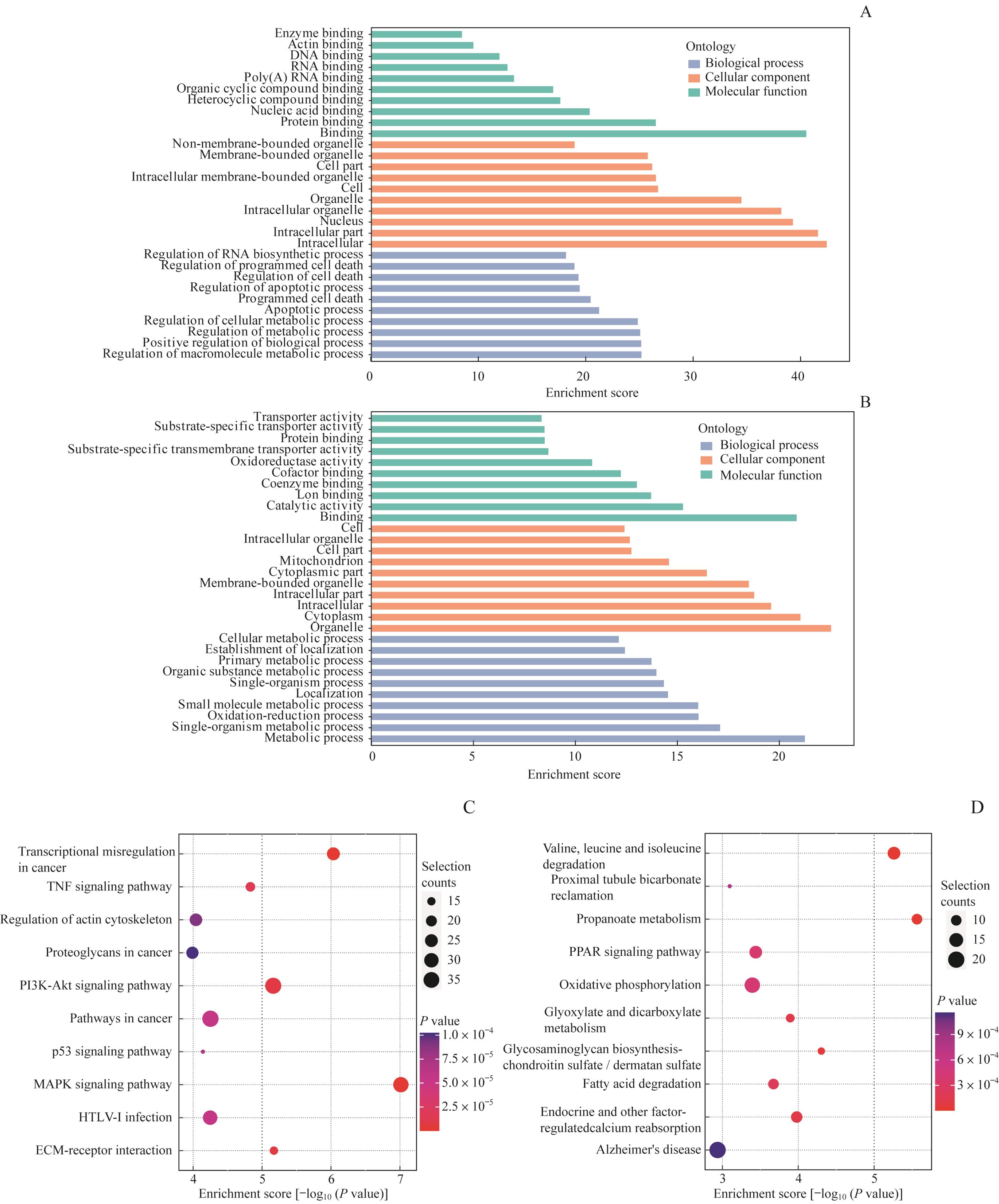
图4 GO与KEGG分析包含DMMS的编码基因Note: A. Major GO terms were enriched for the genes containing up-regulated m6A sites. B. Major GO terms were enriched for the genes containing down-regulated methylated m6A sites. C. Major enriched pathways for the genes containing up-regulated methylated m6A sites. D. Major enriched pathways for the genes containing down-regulated methylated m6A sites.
Fig 4 GO and KEGG analyses of coding genes containing DMMSs
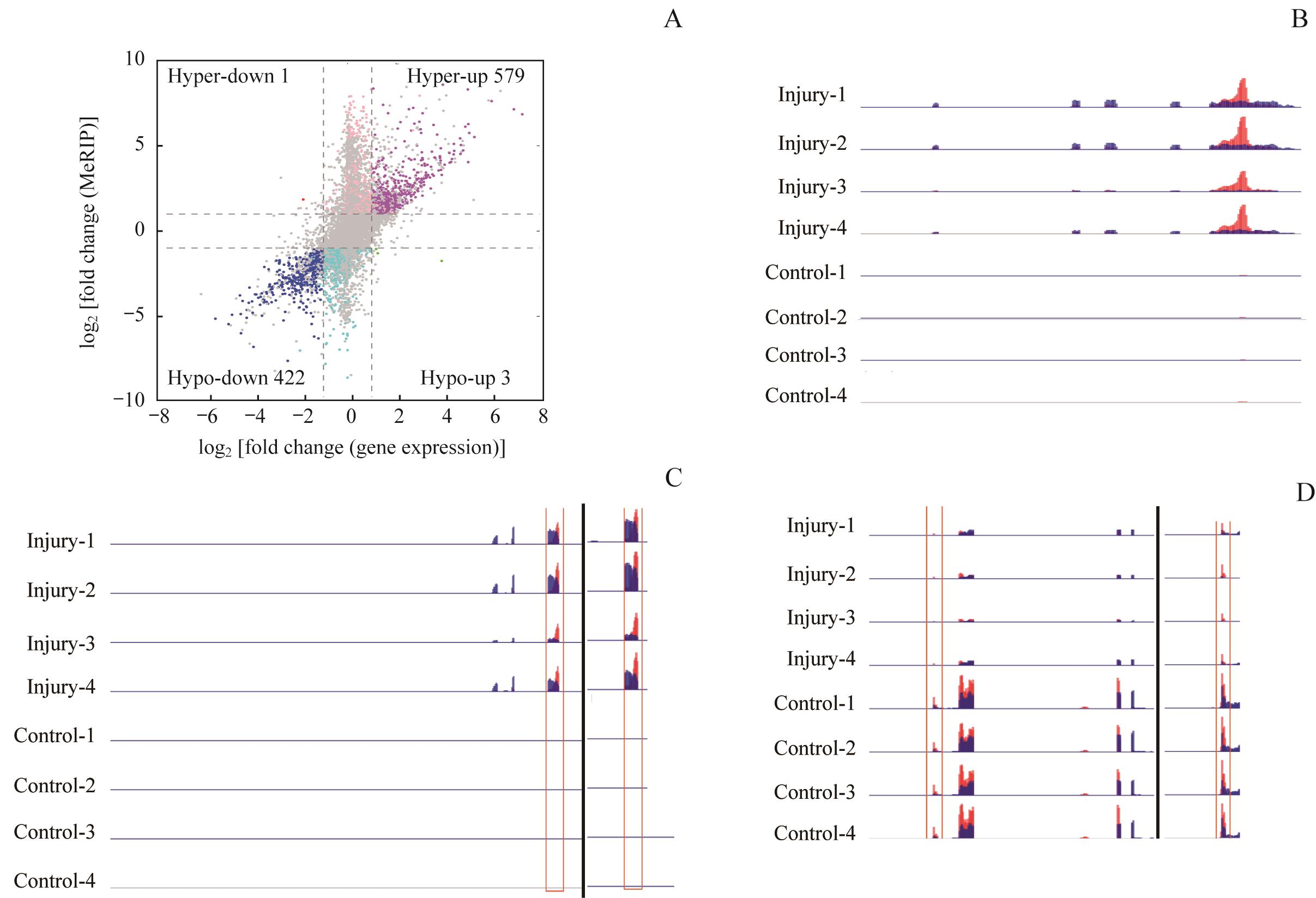
图5 差异甲基化基因和差异表达基因的联合分析Note: A. Four-quadrant graph exhibiting the differentially expressed genes containing DMMS peaks. B. Visualization of m6A-modified gene Fga. C. Visualization of m6A-modified gene Slc12a1. D. Visualization of m6A-modified gene Havcr1.
Fig 5 Conjoint analysis of differentially methylated genes and differentially expressed genes
| 1 | Lebwohl D, Canetta R. Clinical development of platinum complexes in cancer therapy: an historical perspective and an update[J]. Eur J Cancer, 1998, 34(10): 1522-1534. |
| 2 | Ozkok A, Edelstein CL. Pathophysiology of cisplatin-induced acute kidney injury[J]. Biomed Res Int, 2014, 2014: 967826. |
| 3 | Holditch SJ, Brown CN, Lombardi AM, et al. Recent advances in models, mechanisms, biomarkers, and interventions in cisplatin-induced acute kidney injury[J]. Int J Mol Sci, 2019, 20(12): 3011. |
| 4 | Yimit A, Adebali O, Sancar A, et al. Differential damage and repair of DNA-adducts induced by anti-cancer drug cisplatin across mouse organs[J]. Nat Commun, 2019, 10: 309. |
| 5 | Zuk A, Bonventre JV. Acute kidney injury[J]. Annu Rev Med, 2016, 67(1): 293-307. |
| 6 | Sahu BD, Mahesh Kumar J, R.Baicalein Sistla, bioflavonoida, prevents cisplatin-induced acute kidney injury by up-regulating antioxidant defenses and down-regulating the MAPKs and NF-κB pathways[J]. PLoS One, 2015, 10(7): e0134139. |
| 7 | Csepany T, Lin A, Baldick CJ, et al. Sequence specificity of mRNA N6-adenosine methyltransferase[J]. J Biol Chem, 1990, 265(33): 20117-20122. |
| 8 | Lichinchi G, Gao S, Saletore Y, et al. Dynamics of the human and viral m6A RNA methylomes during HIV-1 infection of T cells[J]. Nat Microbiol, 2016, 1: 16011. |
| 9 | Spitale RC, Flynn RA, Zhang QC, et al. Structural imprints in vivo decode RNA regulatory mechanisms[J]. Nature, 2015, 519(7544): 486-490. |
| 10 | Wang Y, Li Y, Toth JI, et al. N6-methyladenosine modification destabilizes developmental regulators in embryonic stem cells[J]. Nat Cell Biol, 2014, 16(2): 191-198. |
| 11 | Wang J, Ishfaq M, Xu L, et al. METTL3/m6A/miRNA-873-5p attenuated oxidative stress and apoptosis in colistin-induced kidney injury by modulating Keap1/Nrf2 pathway[J]. Front Pharmacol, 2019, 10: 517. |
| 12 | Zhou PH, Wu M, Ye CY, et al. Meclofenamic acid promotes cisplatin-induced acute kidney injury by inhibiting fat mass and obesity-associated protein-mediated m6A abrogation in RNA[J]. J Biol Chem, 2019, 294(45): 16908-16917. |
| 13 | Xu Y, Yuan XD, Wu JJ, et al. The N6-methyladenosine mRNA methylase METTL14 promotes renal ischemic reperfusion injury via suppressing YAP1[J]. J Cell Biochem, 2020, 121(1): 524-533. |
| 14 | Leemans JC, Stokman G, Claessen N, et al. Renal-associated TLR2 mediates ischemia/reperfusion injury in the kidney[J]. J Clin Invest, 2005, 115(10): 2894-2903. |
| 15 | Wang L, Feng Z, Wang X, et al. DEGseq: an R package for identifying differentially expressed genes from RNA-seq data[J]. Bioinformatics, 2010, 26(1): 136-138. |
| 16 | Zhou J, Wan J, Gao X, et al. Dynamic m6A mRNA methylation directs translational control of heat shock response[J]. Nature, 2015, 526(7574): 591-594. |
| 17 | Li HB, Tong J, Zhu S, et al. m6A mRNA methylation controls T cell homeostasis by targeting the IL-7/STAT5/SOCS pathways[J]. Nature, 2017, 548(7667): 338-342. |
| 18 | Fry NJ, Law BA, Ilkayeva OR, et al. N6-methyladenosine is required for the hypoxic stabilization of specific mRNAs[J]. RNA, 2017, 23(9): 1444-1455. |
| 19 | Wang Y, Mao J, Wang X, et al. Genome-wide screening of altered m6A-tagged transcript profiles in the hippocampus after traumatic brain injury in mice[J]. Epigenomics, 2019, 11(7): 805-819. |
| 20 | Luo Z, Zhang Z, Tai L, et al. Comprehensive analysis of differences of N6-methyladenosine RNA methylomes between high-fat-fed and normal mouse livers[J]. Epigenomics, 2019, 11(11): 1267-1282. |
| 21 | Dominissini D, Moshitch-Moshkovitz S, Schwartz S, et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq[J]. Nature, 2012, 485(7397): 201-206. |
| 22 | Vilar R, Fish RJ, Casini A, et al. Fibrin(ogen) in human disease: both friend and foe[J]. Haematologica, 2020, 105(2): 284-296. |
| 23 | Meyer KD, Saletore Y, Zumbo P, et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons[J]. Cell, 2012, 149(7): 1635-1646. |
| 24 | Riccioni G, Gammone M, Currenti W, et al. Effectiveness and safety of dietetic supplementation of a new nutraceutical on lipid profile and serum inflammation biomarkers in hypercholesterolemic patients[J]. Molecules, 2018, 23(5): 1168. |
| 25 | Markadieu N, Delpire E. Physiology and pathophysiology of SLC12A1/2 transporters[J]. Pflugers Arch, 2014, 466(1): 91-105. |
| 26 | Martini F, Cecconi N, Paolicchi A, et al. Interference of monoclonal gammopathy with fibrinogen assay producing spurious dysfibrinogenemia[J]. TH Open, 2019, 3(1): e64-e66. |
| 27 | Hao S, Hao M, Ferreri NR. Renal-specific silencing of TNF (tumor necrosis factor) unmasks salt-dependent increases in blood pressure via an NKCC2A (Na+-K+-2Cl- cotransporter isoform A)-dependent mechanism[J]. Hypertension, 2018, 71(6): 1117-1125. |
| 28 | Zdziechowska M, Gluba-Brzózka A, Franczyk B, et al. Biochemical markers in the prediction of contrast-induced acute kidney injury[J]. Curr Med Chem, 2021, 28(6): 1234-1250. |
| 29 | Lippi I, Perondi F, Meucci V, et al. Clinical utility of urine kidney injury molecule-1 (KIM-1) and gamma-glutamyl transferase (GGT) in the diagnosis of canine acute kidney injury[J]. Vet Res Commun, 2018, 42(2): 95-100. |
| 30 | Yang L, Brooks CR, Xiao S, et al. KIM-1-mediated phagocytosis reduces acute injury to the kidney[J]. J Clin Invest, 2015, 125(4): 1620-1636. |
| 31 | Song J, Yu J, Prayogo GW, et al. Understanding kidney injury molecule 1: a novel immune factor in kidney pathophysiology[J]. Am J Transl Res, 2019, 11(3): 1219-1229. |
| [1] | 吴望舒, 王旻洲, 宋阿会, 赵冰茹, 鲁嘉越, 洪文凯, 顾乐怡, 谢可炜, 陆任华. 复方氨基酸胶囊治疗维持性血液透析患者营养不良及钙磷代谢障碍的有效性和安全性[J]. 上海交通大学学报(医学版), 2024, 44(8): 1023-1029. |
| [2] | 魏珊, 纪鸥洋, 陈志豪, 黄泽慧, 李璞, 方均燕, 刘英莉. 维持性透析患者安全用药知信行现状调查及相关因素分析[J]. 上海交通大学学报(医学版), 2023, 43(1): 88-94. |
| [3] | 王亚琨, 许佳瑞, 吴茜茜, 张晓华, 朱迎春, 白寿军. 医养结合综合干预对上海郊区老年慢性肾脏病患者生活质量和精神状态的影响[J]. 上海交通大学学报(医学版), 2022, 42(7): 904-910. |
| [4] | 黄泽慧, 胡春, 李璞, 张春丽, 方均燕, 宋阿会, 魏珊, 纪鸥洋, 佟琰, 邓海, 刘英莉. 居家腹膜透析患者自我管理量表的编制及信度、效度分析研究[J]. 上海交通大学学报(医学版), 2021, 41(7): 942-948. |
| [5] | 周悦玲,丁 巍,艾红兰,卢建新,丁 峰,胡 春. 维持性血液透析的终末期肾病患者脑结构性异常及认知功能分析[J]. 上海交通大学学报(医学版), 2020, 40(7): 962-967. |
| [6] | 王丽 1, 2,章倩莹 1,林涛 1,徐天 1,黄晓敏 1,张春燕 1,徐耀文 1,吴珮 1,陈楠 1,任红 1,谢静远 1. 腹膜炎是腹膜透析患者全因死亡及心血管死亡的独立危险因素[J]. 上海交通大学学报(医学版), 2019, 39(9): 1024-. |
| [7] | 周培慧,王丽. 白细胞介素 -27在顺铂所致急性肾损伤中的高表达及对细胞 凋亡的抑制作用[J]. 上海交通大学学报(医学版), 2019, 39(4): 372-. |
| [8] | 苏新玙 *,刘苗 *,俞赞喆,严豪,李振元,张贺,袁江姿,倪兆慧,方炜. 临床常用指标在腹膜透析患者容量状态评估中的价值[J]. 上海交通大学学报(医学版), 2018, 38(8): 910-. |
| [9] | 庞慧华,章海芬,车妙琳,俞赞喆,林星辉,朱铭力,陆任华,方妮娜,倪兆慧,顾乐怡. 维持性血液透析患者容量状况的评估[J]. 上海交通大学学报(医学版), 2018, 38(5): 524-. |
| [10] | 付玉玲1,胡坤1,沈艳萍1,施鑫2,乔青1. 持续非卧床腹膜透析患者成纤维生长因子 -23及可溶性klotho蛋白水平与心脏瓣膜钙化的关系[J]. 上海交通大学学报(医学版), 2018, 38(5): 541-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||