
上海交通大学学报(医学版) ›› 2026, Vol. 46 ›› Issue (3): 377-384.doi: 10.3969/j.issn.1674-8115.2026.03.012
• 综述 • 上一篇
徐蕊1,2,3,4, 郭嘉城2,3,4, 谢诗瑶2,3,4, 韩德恒2,3,4, 岳修勤1( )
)
收稿日期:2025-09-07
接受日期:2025-11-27
出版日期:2026-03-28
发布日期:2026-03-30
通讯作者:
岳修勤,主任医师,博士;电子信箱:xiuqinyue@163.com。基金资助:
Xu Rui1,2,3,4, Guo Jiacheng2,3,4, Xie Shiyao2,3,4, Han Deheng2,3,4, Yue Xiuqin1( )
)
Received:2025-09-07
Accepted:2025-11-27
Online:2026-03-28
Published:2026-03-30
Contact:
Yue Xiuqin, E-mail: xiuqinyue@163.com.Supported by:摘要:
心脏纤维化是多种心血管疾病常见且关键的病理环节,显著阻碍心功能恢复并影响远期预后。传统药物如肾素-血管紧张素-醛固酮系统抑制剂虽有一定抗心脏纤维化作用,但其靶向性不足、作用机制相对局限,难以实现对心脏纤维化核心通路的精准干预。近年来,纳米材料凭借其独特的尺寸效应、可调控的表面性质及良好的生物相容性,在心脏纤维化的精准治疗与组织修复中展现出巨大潜力。该文综述纳米材料在心脏纤维化修复中的最新进展,重点阐述其精准递送、智能控释、功能仿生以及多功能复合集成等策略,剖析纳米材料如何从分子层面调控关键信号通路,在细胞层面调控成纤维细胞、免疫细胞及内皮细胞等功能,进而促进组织层面的结构重建与功能恢复,实现从精准干预到组织重塑的系统性修复。该文还探讨了纳米材料在心血管疾病领域临床转化的现状与挑战,包括其生物安全性、规模化生产及跨物种转化等问题,并展望其未来发展方向,旨在为开发高效、安全的抗心脏纤维化纳米治疗策略提供理论参考与实践指引。
中图分类号:
徐蕊, 郭嘉城, 谢诗瑶, 韩德恒, 岳修勤. 纳米材料应用于心脏纤维化修复的研究进展:从精准治疗到组织重塑[J]. 上海交通大学学报(医学版), 2026, 46(3): 377-384.
Xu Rui, Guo Jiacheng, Xie Shiyao, Han Deheng, Yue Xiuqin. Research advances in nanomaterials in cardiac fibrosis repair: from precision therapy to tissue remodeling[J]. Journal of Shanghai Jiao Tong University (Medical Science), 2026, 46(3): 377-384.
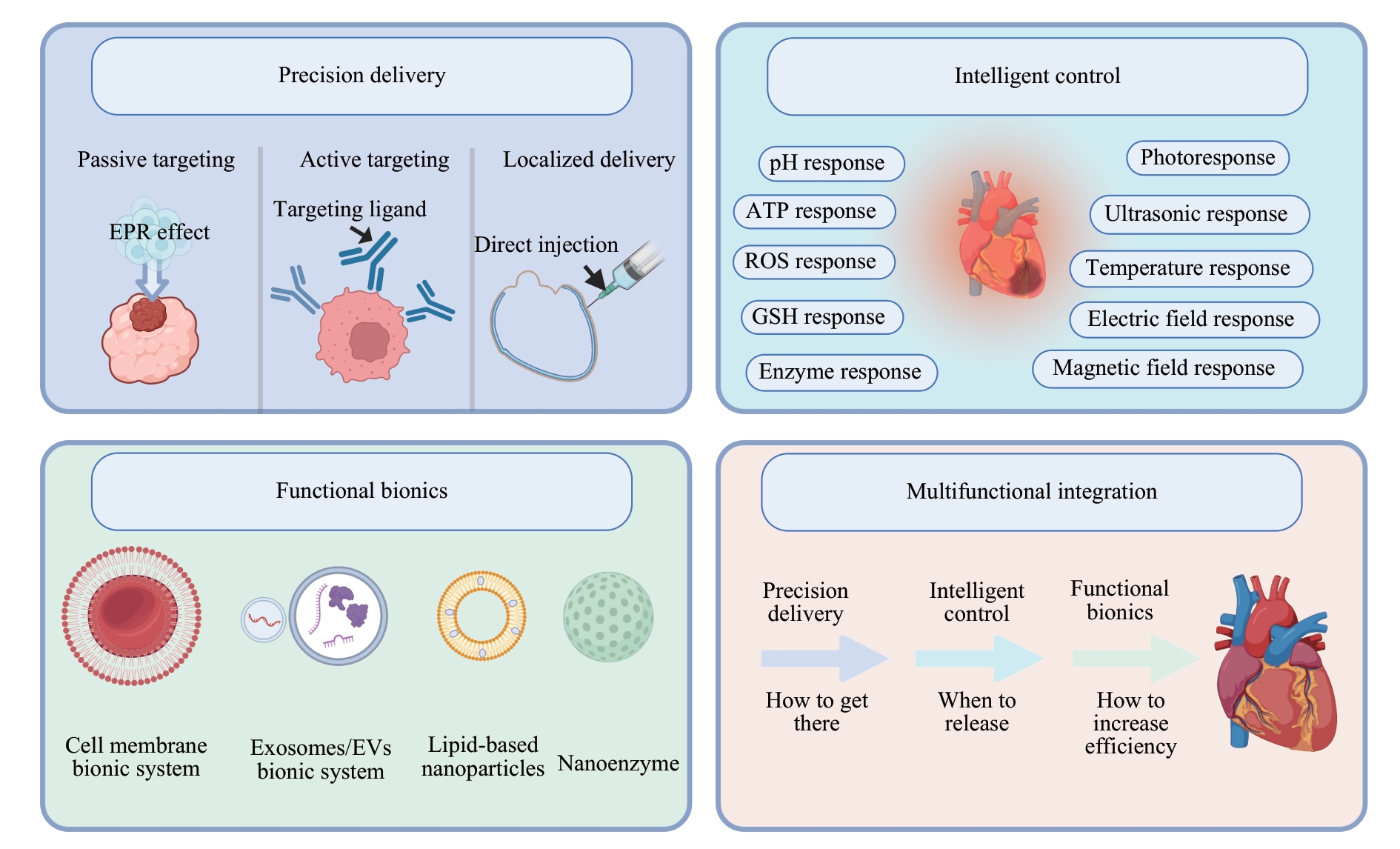
图1 心脏纤维化纳米治疗的多维策略示意图(BioRender绘制)Note: EPR—enhanced permeability and retention; ATP—adenosine triphosphate; ROS—reactive oxygen species; GSH—glutathione; EVs—extracellular vesicles.
Fig 1 Schematic diagram of multidimensional strategies for nanotherapy for cardiac fibrosis (created with BioRender)
| Material type | Representative system/material | Research model | Phenotypic effect | Off-target degree | Clinical trial phase |
|---|---|---|---|---|---|
| Inorganic nanoparticle | Nano-SiO₂/TiO₂/Fe₂O₃, et al[ | In vitro, small animal | Imaging enhancement, photothermal therapy, drug carrier | High | Preclinical/limited clinical; Fe₂O₃ contrast agent has been withdrawn |
| Organic Nanoparticle | |||||
| Lipid nanoparticle | Lipid nanoparticle[ | In vitro, small animal, large animal, human trial | Nucleic acid delivery (mRNA/siRNA), gene editing | Low-medium | Phase Ⅰ/Ⅱ (primary) |
| Liposome | Functionalized liposome (e.g., pH/ROS-responsive)[ | In vitro, small animal, human trial | Gene delivery, small-molecule drug delivery, toxicity reduction | Low | Phase Ⅰ/Ⅱ (primary); multiple liposomal drugs are marketed (e.g., doxorubicin, amphotericin B liposomes) |
| Exosome/EVs | Stem cell-derived exosome, engineered exosome[ | In vitro, small animal, large animal, human trial | Natural targeting, anti-inflammatory, tissue repair | High | Phase Ⅰ/Ⅱ (primary) |
| PEG nanocarrier | |||||
| Polymeric micelle | PLA-PEG micelle[ | In vitro, small animal | RNA encapsulation and delivery | Low-medium | Preclinical; Genexol-PM® is marketed in several countries |
| Polymeric nanoparticle | PLGA-PEG nanoparticle[ | In vitro, small animal | Long-acting sustained release | Low | Preclinical |
| Other organic nanoparticle | Protein nanoparticle[ | In vitro, small animal | Sustained-release drug carrier | Low | Phase Ⅰ/Ⅱ (primary); albumin nanoparticles (Abraxane®) are marketed (for cancer) |
| Organic-inorganic hybrid nanoparticle | Protein-nanoparticle hybrid material[ | In vitro, small animal | Synergistic therapy | Medium | Preclinical |
表1 代表性纳米材料在心脏纤维化治疗中的临床前研究及临床转化概况
Tab 1 Overview of representative nanomaterials for cardiac fibrosis therapy: preclinical studies and clinical translation
| Material type | Representative system/material | Research model | Phenotypic effect | Off-target degree | Clinical trial phase |
|---|---|---|---|---|---|
| Inorganic nanoparticle | Nano-SiO₂/TiO₂/Fe₂O₃, et al[ | In vitro, small animal | Imaging enhancement, photothermal therapy, drug carrier | High | Preclinical/limited clinical; Fe₂O₃ contrast agent has been withdrawn |
| Organic Nanoparticle | |||||
| Lipid nanoparticle | Lipid nanoparticle[ | In vitro, small animal, large animal, human trial | Nucleic acid delivery (mRNA/siRNA), gene editing | Low-medium | Phase Ⅰ/Ⅱ (primary) |
| Liposome | Functionalized liposome (e.g., pH/ROS-responsive)[ | In vitro, small animal, human trial | Gene delivery, small-molecule drug delivery, toxicity reduction | Low | Phase Ⅰ/Ⅱ (primary); multiple liposomal drugs are marketed (e.g., doxorubicin, amphotericin B liposomes) |
| Exosome/EVs | Stem cell-derived exosome, engineered exosome[ | In vitro, small animal, large animal, human trial | Natural targeting, anti-inflammatory, tissue repair | High | Phase Ⅰ/Ⅱ (primary) |
| PEG nanocarrier | |||||
| Polymeric micelle | PLA-PEG micelle[ | In vitro, small animal | RNA encapsulation and delivery | Low-medium | Preclinical; Genexol-PM® is marketed in several countries |
| Polymeric nanoparticle | PLGA-PEG nanoparticle[ | In vitro, small animal | Long-acting sustained release | Low | Preclinical |
| Other organic nanoparticle | Protein nanoparticle[ | In vitro, small animal | Sustained-release drug carrier | Low | Phase Ⅰ/Ⅱ (primary); albumin nanoparticles (Abraxane®) are marketed (for cancer) |
| Organic-inorganic hybrid nanoparticle | Protein-nanoparticle hybrid material[ | In vitro, small animal | Synergistic therapy | Medium | Preclinical |
| [1] | Zhang Y Z, Lin X Y, Chu Y, et al. Dapagliflozin: a sodium-glucose cotransporter 2 inhibitor, attenuates angiotensin Ⅱ-induced cardiac fibrotic remodeling by regulating TGFβ1/Smad signaling[J]. Cardiovasc Diabetol, 2021, 20(1): 121. |
| [2] | Chen X F, Yang Q, Bai W L, et al. Dapagliflozin attenuates myocardial fibrosis by inhibiting the TGF-β1/Smad signaling pathway in a normoglycemic rabbit model of chronic heart failure[J]. Front Pharmacol, 2022, 13: 873108. |
| [3] | Chinnappa S, Maqbool A, Viswambharan H, et al. Beta blockade prevents cardiac morphological and molecular remodelling in experimental uremia[J]. Int J Mol Sci, 2023, 25(1): 373. |
| [4] | Ibrahim W S, Ahmed H M S, Mahmoud A A A, et al. Propranolol and low-dose isoproterenol ameliorate insulin resistance, enhance β-arrestin2 signaling, and reduce cardiac remodeling in high-fructose, high-fat diet-fed mice: comparative study with metformin[J]. Life Sci, 2021, 286: 120055. |
| [5] | Cunningham J W, Claggett B L, O′Meara E, et al. Effect of sacubitril/valsartan on biomarkers of extracellular matrix regulation in patients with HFpEF[J]. J Am Coll Cardiol, 2020, 76(5): 503-514. |
| [6] | Frangogiannis N G. Cardiac fibrosis[J]. Cardiovasc Res, 2021, 117(6): 1450-1488. |
| [7] | Tie Y, Tang F, Peng D D, et al. TGF-β signal transduction: biology, function and therapy for diseases[J]. Mol Biomed, 2022, 3(1): 45. |
| [8] | Zhang T T, Lei Q Q, He J, et al. Bestrophin3 deficiency in vascular smooth muscle cells activates MEKK2/3-MAPK signaling to trigger spontaneous aortic dissection[J]. Circulation, 2023, 148(7): 589-606. |
| [9] | Cheng T H, Cheng P Y, Shih N L, et al. Involvement of reactive oxygen species in angiotensin Ⅱ-induced endothelin-1 gene expression in rat cardiac fibroblasts[J]. J Am Coll Cardiol, 2003, 42(10): 1845-1854. |
| [10] | Bretherton R, Bugg D, Olszewski E, et al. Regulators of cardiac fibroblast cell state[J]. Matrix Biol, 2020, 91/92: 117-135. |
| [11] | Nayakanti S R, Friedrich A, Sarode P, et al. Targeting Wnt-β-catenin-FOSL signaling ameliorates right ventricular remodeling[J]. Circ Res, 2023, 132(11): 1468-1485. |
| [12] | Blyszczuk P, Müller-Edenborn B, Valenta T, et al. Transforming growth factor-β-dependent Wnt secretion controls myofibroblast formation and myocardial fibrosis progression in experimental autoimmune myocarditis[J]. Eur Heart J, 2017, 38(18): 1413-1425. |
| [13] | Cho S, Rhee S, Madl C M, et al. Selective inhibition of stromal mechanosensing suppresses cardiac fibrosis[J]. Nature, 2025, 642(8068): 766-775. |
| [14] | Jiang H, Yang J J, Li T, et al. JAK/STAT3 signaling in cardiac fibrosis: a promising therapeutic target[J]. Front Pharmacol, 2024, 15: 1336102. |
| [15] | Shi J L, Xiao P L, Liu X L, et al. Notch3 modulates cardiac fibroblast proliferation, apoptosis, and fibroblast to myofibroblast transition via negative regulation of the RhoA/ROCK/HIF1α axis[J]. Front Physiol, 2020, 11: 669. |
| [16] | Senesi G, Lodrini A M, Mohammed S, et al. miR-24-3p secreted as extracellular vesicle cargo by cardiomyocytes inhibits fibrosis in human cardiac microtissues[J]. Cardiovasc Res, 2025, 121(1): 143-156. |
| [17] | Li G Q, Shao Y H, Guo H C, et al. MicroRNA-27b-3p down-regulates FGF1 and aggravates pathological cardiac remodelling[J]. Cardiovasc Res, 2022, 118(9): 2139-2151. |
| [18] | Zhou J T, Tian G E, Quan Y, et al. The long noncoding RNA THBS1-AS1 promotes cardiac fibroblast activation in cardiac fibrosis by regulating TGFBR1[J]. JCI Insight, 2023, 8(6): e160745. |
| [19] | Wang X H, Chen T, Chen S Z, et al. STING aggravates ferroptosis-dependent myocardial ischemia-reperfusion injury by targeting GPX4 for autophagic degradation[J]. Signal Transduct Target Ther, 2025, 10(1): 136. |
| [20] | Horwitz L D, Kaufman D, Keller M W, et al. Time course of coronary endothelial healing after injury due to ischemia and reperfusion[J]. Circulation, 1994, 90(5): 2439-2447. |
| [21] | Prajnamitra R P, Chen H C, Lin C J, et al. Nanotechnology approaches in tackling cardiovascular diseases[J]. Molecules, 2019, 24(10): 2017. |
| [22] | Wen J X, Guan Y, Niu H, et al. Targeting cardiac resident CCR2+ macrophage-secreted MCP-1 to attenuate inflammation after myocardial infarction[J]. Acta Biomater, 2024: S1742-S7061(24)00469-0. |
| [23] | Chen W S, Liu L J, Tang M, et al. Type Ⅰ collagen-targeted liposome delivery of Serca2a modulates myocardium calcium homeostasis and reduces cardiac fibrosis induced by myocardial infarction[J]. Mater Today Bio, 2024, 28: 101162. |
| [24] | Liu J, Zhuang T, Pi J J, et al. Endothelial forkhead box transcription factor P1 regulates pathological cardiac remodeling through transforming growth factor-β1-endothelin-1 signal pathway[J]. Circulation, 2019, 140(8): 665-680. |
| [25] | Zhu D S, Liu S, Huang K, et al. Intrapericardial long non-coding RNA—Tcf21 antisense RNA inducing demethylation administration promotes cardiac repair[J]. Eur Heart J, 2023, 44(19): 1748-1760. |
| [26] | Liang X Y, Zhang Y, Wang C D, et al. ROS-responsive death receptor 5 fusion protein nano-delivery system enhances myocardial ischemia-reperfusion injury protection[J]. Mater Today Bio, 2025, 32: 101899. |
| [27] | Sullivan H L, Liang Y F, Worthington K, et al. Enzyme-responsive nanoparticles for the targeted delivery of an MMP inhibitor to acute myocardial infarction[J]. Biomacromolecules, 2023, 24(11): 4695-4704. |
| [28] | Fatima M, Almalki W H, Khan T, et al. Harnessing the power of stimuli-responsive nanoparticles as an effective therapeutic drug delivery system[J]. Adv Mater, 2024, 36(24): e2312939. |
| [29] | Zhao X L, Qin Y Z, Li B W, et al. Genetically engineered biomimetic ATP-responsive nanozyme for the treatment of cardiac fibrosis[J]. J Nanobiotechnology, 2025, 23(1): 10. |
| [30] | Ji X Q, Meng Y B, Wang Q Y, et al. Cysteine-based redox-responsive nanoparticles for fibroblast-targeted drug delivery in the treatment of myocardial infarction[J]. ACS Nano, 2023, 17(6): 5421-5434. |
| [31] | Dong Y X, Wang H B, Zhang Y L, et al. NIR-Ⅱ light based combinatorial management of hypertrophic scar by inducing autophagy in fibroblasts[J]. J Nanobiotechnology, 2024, 22(1): 625. |
| [32] | Yang J L, He J, Yue T, et al. The clinical prospects and challenges of photothermal nanomaterials in myocardium recovery after myocardial infarction[J]. Front Bioeng Biotechnol, 2024, 12: 1491581. |
| [33] | Wang X Y, Bai R R. Advances in smart delivery of magnetic field-targeted drugs in cardiovascular diseases[J]. Drug Deliv, 2023, 30(1): 2256495. |
| [34] | Zhang L H, Bei Z W, Li T, et al. An injectable conductive hydrogel with dual responsive release of rosmarinic acid improves cardiac function and promotes repair after myocardial infarction[J]. Bioact Mater, 2023, 29: 132-150. |
| [35] | He M Q, Li Y, Zhu D S, et al. Stromal-platelet membrane-inspired nanoparticles (SPIN) for targeted heart repair[J]. Bioact Mater, 2025, 53: 45-57. |
| [36] | Chen Q, Yu Y, Tong L, et al. Targeted delivery of TAPI-1 via biomimetic nanoparticles ameliorates post-infarct left ventricle function and remodelling[J]. Cardiovasc Res, 2025, 121(5): 760-774. |
| [37] | Li Y Y, Yu J Q, Cheng C, et al. Platelet and erythrocyte membranes coassembled biomimetic nanoparticles for heart failure treatment[J]. ACS Nano, 2024, 18(39): 26614-26630. |
| [38] | Lu H, Jiang Y H, Luo R, et al. Engineered hybrid cell membrane nanosystems for treating cardiovascular diseases[J]. Mater Today Bio, 2025, 33: 101992. |
| [39] | Yu T T, Xu Q X, Chen X, et al. Biomimetic nanomaterials in myocardial infarction treatment: harnessing bionic strategies for advanced therapeutics[J]. Mater Today Bio, 2024, 25: 100957. |
| [40] | Wang X, Chen Y H, Zhao Z N, et al. Engineered exosomes with ischemic myocardium-targeting peptide for targeted therapy in myocardial infarction[J]. J Am Heart Assoc, 2018, 7(15): e008737. |
| [41] | Ihnatsyeu-Kachan A, Saichuk A, Sharko O, et al. Biomimetic high-density lipoprotein nanoparticles for the delivery of nucleic acid-based therapeutics[J]. Biotechnol Adv, 2025, 83: 108606. |
| [42] | Gong Y X, Xiao Y, Zhao C Y, et al. Ultrasmall PtIr bimetallic nanozyme treats myocardial infarction via ischemic/inflammatory cardiac microenvironment remodeling[J]. ACS Nano, 2025, 19(14): 13723-13739. |
| [43] | Gu Z Y, Liu X L, Qi Z, et al. An antioxidant nanozyme for targeted cardiac fibrosis therapy post myocardial infarction[J]. J Nanobiotechnology, 2024, 22(1): 760. |
| [44] | Li F Y, Liu D Z, Liu M, et al. Tregs biomimetic nanoparticle to reprogram inflammatory and redox microenvironment in infarct tissue to treat myocardial ischemia reperfusion injury in mice[J]. J Nanobiotechnology, 2022, 20(1): 251. |
| [45] | Zhu K, Wang K, Zhang R T, et al. Iron chelators loaded on myocardiocyte mitochondria-targeted nanozyme system for treating myocardial ischemia-reperfusion injury in mouse models[J]. J Nanobiotechnology, 2025, 23(1): 112. |
| [46] | Liu Z J, Chen C, Zhang Y L, et al. Legumain in situ engineering promotes efferocytosis of CAR macrophage to treat cardiac fibrosis[J]. Adv Mater, 2025, 37(27): e2417831. |
| [47] | Morsink M, Severino P, Luna-Ceron E, et al. Effects of electrically conductive nano-biomaterials on regulating cardiomyocyte behavior for cardiac repair and regeneration[J]. Acta Biomater, 2022, 139: 141-156. |
| [48] | Chen C X, Ma J J, Duan S J, et al. Mitigation of ischemia/reperfusion injury via selenium nanoparticles: suppression of STAT1 to inhibit cardiomyocyte oxidative stress and inflammation[J]. Biomaterials, 2025, 318: 123119. |
| [49] | Wang Y, Li C F, Zhao R Z, et al. CircUbe3a from M2 macrophage-derived small extracellular vesicles mediates myocardial fibrosis after acute myocardial infarction[J]. Theranostics, 2021, 11(13): 6315-6333. |
| [50] | Chen S Q, Wang K, Fan Z F, et al. Modulation of anti-cardiac fibrosis immune responses by changing M2 macrophages into M1 macrophages[J]. Mol Med, 2024, 30(1): 88. |
| [51] | Rurik J G, Tombácz I, Yadegari A, et al. CAR T cells produced in vivo to treat cardiac injury[J]. Science, 2022, 375(6576): 91-96. |
| [52] | Esmaeili H, Patino-Guerrero A, Nelson R A Jr, et al. Engineered gold and silica nanoparticle-incorporated hydrogel scaffolds for human stem cell-derived cardiac tissue engineering[J]. ACS Biomater Sci Eng, 2024, 10(4): 2351-2366. |
| [53] | Cheng N L, Luo Q, Yang Y Q, et al. Injectable pH responsive conductive hydrogel for intelligent delivery of metformin and exosomes to enhance cardiac repair after myocardial ischemia-reperfusion injury[J]. Adv Sci (Weinh), 2025, 12(24): e2410590. |
| [54] | Yajima S, Lee S H, Yang J K, et al. Stromal cell-derived factor-encapsulated nanoparticles target ischemic myocardium and attenuate myocardial injury via proangiogenic effects[J]. Biomaterials, 2025, 318: 123167. |
| [55] | Cui X L, Guo J C, Yuan P Y, et al. Bioderived nanoparticles for cardiac repair[J]. ACS Nano, 2024, 18(36): 24622-24649. |
| [56] | Femminò S, D′Ascenzo F, Ravera F, et al. Percutaneous coronary intervention (PCI) reprograms circulating extracellular vesicles from ACS patients impairing their cardio-protective properties[J]. Int J Mol Sci, 2021, 22(19): 10270. |
| [57] | Yang L, Mei J Q, Qiao F, et al. Randomized controlled trial assessing the efficacy and safety of a liposomal carrier for low-dose dual antiplatelet therapy (clopidogrel and aspirin) in coronary heart disease patients[J]. Clin Ther, 2025, 47(5): 339-346. |
| [58] | Sun B X, Liu S W, Hao R B, et al. RGD-PEG-PLA delivers miR-133 to infarct lesions of acute myocardial infarction model rats for cardiac protection[J]. Pharmaceutics, 2020, 12(6): 575. |
| [59] | Zhu K, Jiang D W, Wang K, et al. Conductive nanocomposite hydrogel and mesenchymal stem cells for the treatment of myocardial infarction and non-invasive monitoring via PET/CT[J]. J Nanobiotechnology, 2022, 20(1): 211. |
| [60] | Sharma V, Dash S K, Manhas A, et al. Injectable hydrogel for co-delivery of 5-azacytidine in zein protein nanoparticles with stem cells for cardiac function restoration[J]. Int J Pharm, 2021, 603: 120673. |
| [1] | 徐彤彤, 阮慧瞳. 负载白细胞介素-4的水凝胶微球调控卒中后免疫微环境对神经功能修复的影响[J]. 上海交通大学学报(医学版), 2025, 45(9): 1161-1170. |
| [2] | 鲁佳艺, 刘锦喆, 郭尚春, 陶诗聪. 纳米材料通过降低活性氧水平促进骨组织再生的研究进展[J]. 上海交通大学学报(医学版), 2025, 45(4): 487-492. |
| [3] | 陈亮, 吴彪, 袁良喜. 基于纳米材料的递送系统在动脉粥样硬化诊疗中的应用进展[J]. 上海交通大学学报(医学版), 2025, 45(12): 1687-1693. |
| [4] | 王昊, 王然, 巴乾. 食品中二氧化钛纳米材料对消化道组织及肠道微生物群影响的研究进展[J]. 上海交通大学学报(医学版), 2022, 42(2): 225-229. |
| [5] | 陈 蓦,陈 俊,陈世益. 基于近红外二区荧光纳米探针的活体光学成像技术在生物医学应用的研究进展[J]. 上海交通大学学报(医学版), 2020, 40(4): 530-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||