
上海交通大学学报(医学版) ›› 2026, Vol. 46 ›› Issue (4): 415-426.doi: 10.3969/j.issn.1674-8115.2026.04.001
• 前沿述评 •
王震1, 张楠1, 沈艺冰2, 李丹2, 彭程1( ), 顾志冬3,4,5(
), 顾志冬3,4,5( ), 李斌1,2,3(
), 李斌1,2,3( )
)
收稿日期:2025-12-02
接受日期:2026-01-21
出版日期:2026-04-17
发布日期:2026-04-17
通讯作者:
李 斌,教授,博士;电子信箱:binli@shsmu.edu.cn作者简介:第一联系人:为共同第一作者(co-first authors)。
基金资助:
Wang Zhen1, Zhang Nan1, Shen Yibing2, Li Dan2, Peng Cheng1( ), Gu Zhidong3,4,5(
), Gu Zhidong3,4,5( ), Li Bin1,2,3(
), Li Bin1,2,3( )
)
Received:2025-12-02
Accepted:2026-01-21
Online:2026-04-17
Published:2026-04-17
Contact:
Li Bin, E-mail: binli@shsmu.edu.cnSupported by:摘要:
免疫耐受是维持机体免疫系统稳态的核心基石,一旦这一精密调控机制发生紊乱,将直接诱发类风湿关节炎、系统性红斑狼疮等自身免疫病,以及肿瘤细胞免疫逃逸、器官移植排斥等一系列表现,严重威胁人类生命健康。2025年诺贝尔生理学/医学奖被授予玛丽·E·布伦科(Mary E. Brunkow)、弗雷德·拉姆斯德尔(Fred Ramsdell)与坂口志文(Shimon Sakaguchi),以表彰他们在外周免疫耐受领域的开创性贡献——成功鉴定CD4+CD25+FOXP3+调节性T细胞(regulatory T cell,Treg)及其核心调控基因——叉头框蛋白P3(forkhead box P3,FOXP3),建立了“FOXP3-Treg-免疫耐受”的核心理论框架,为后续相关研究的开展奠定了坚实基础。该综述系统梳理Treg与FOXP3研究的历史脉络,从早期对免疫耐受现象的初步探索,到关键细胞亚群与调控基因的鉴定,清晰呈现领域发展的演进历程;重点聚焦近年来的关键进展,全面涵盖Treg亚型的精细分类与发育特征、FOXP3调控网络的多维度拓展、Treg在多种疾病中的功能机制解析及临床转化的突破性成果等核心内容,最后围绕Treg疗法的临床转化瓶颈对未来发展方向进行展望。
中图分类号:
王震, 张楠, 沈艺冰, 李丹, 彭程, 顾志冬, 李斌. 调节性T细胞基础机制研究突破和临床转化前景[J]. 上海交通大学学报(医学版), 2026, 46(4): 415-426.
Wang Zhen, Zhang Nan, Shen Yibing, Li Dan, Peng Cheng, Gu Zhidong, Li Bin. Breakthroughs in basic mechanism research and clinical translation prospects of regulatory T cells[J]. Journal of Shanghai Jiao Tong University (Medical Science), 2026, 46(4): 415-426.
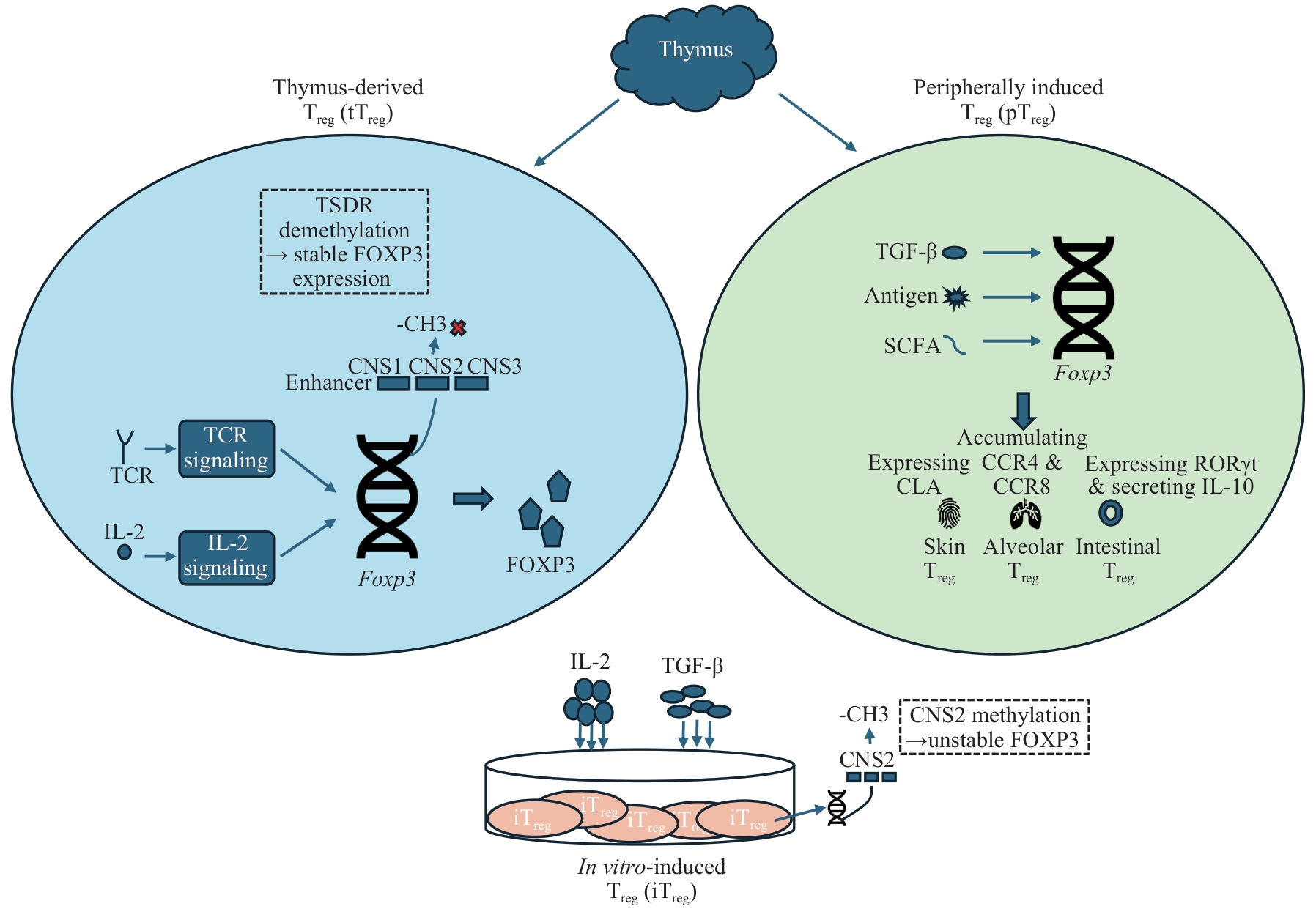
图1 Treg 亚型和发育特征Note: This figure illustrates the subclassification of Treg and associated molecular mechanisms, including tTreg, pTreg, and iTreg. tTreg develop in the thymus under T cell receptor (TCR) and IL-2 signaling, with stable FOXP3 expression maintained by demethylation of conserved non-coding sequence 2 (CNS2). pTreg arise from naive T cells in peripheral tissues in response to transforming growth factor-β (TGF-β), antigen, and short-chain fatty acids (SCFAs), leading to transient FOXP3 expression. iTreg are generated using IL-2 and TGF-β, but exhibit unstable FOXP3 expression due to CNS2 methylation. The distinct epigenetic and environmental cues shape Treg stability and functions across different developmental pathways. TSDR—Treg-specific demethylated region; CLA—cutaneous lymphocyte-associated antigen; CCR4—C-C chemokine receptor 4; RORγt—RAR-related orphan receptor γt.
Fig 1 Subtypes and developmental characteristics of Treg
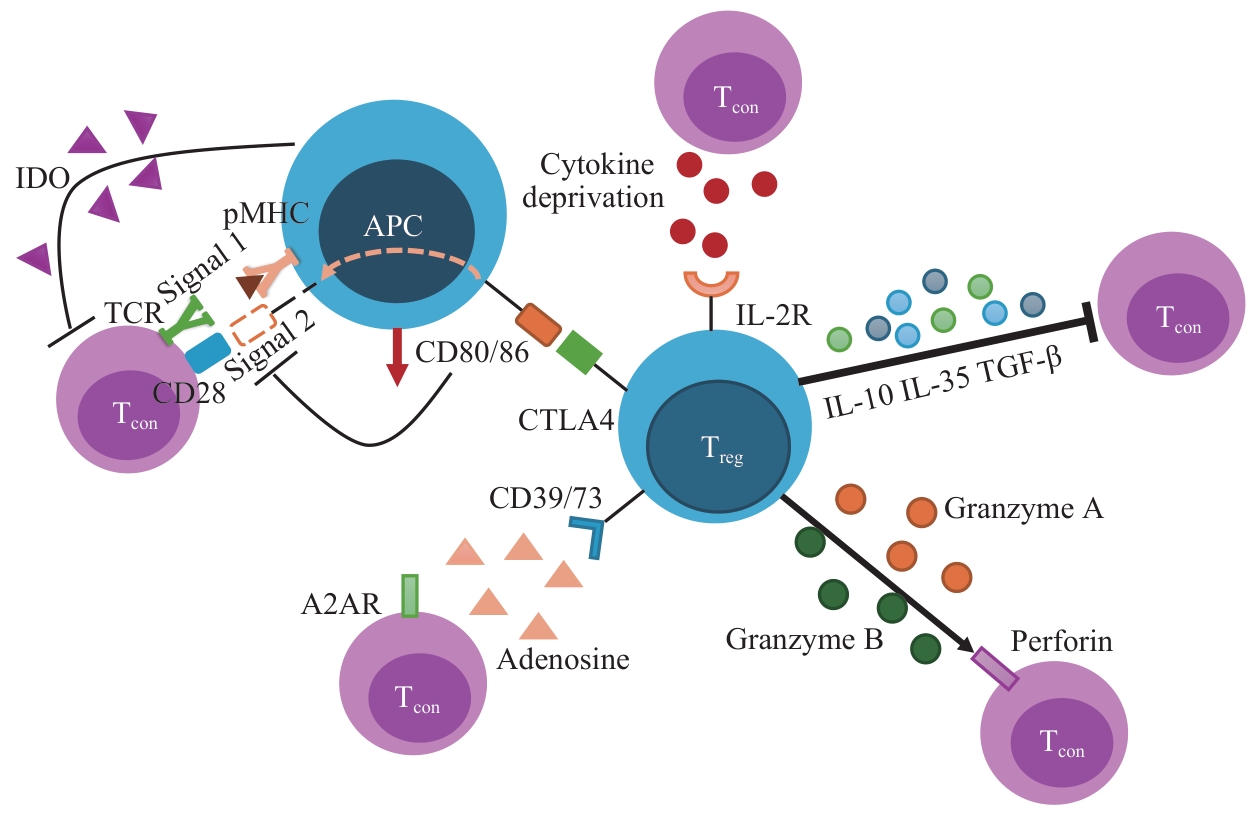
图2 Treg 多重免疫抑制机制Note: This diagram illustrates the key immunosuppressive mechanisms of Treg. Treg exert immunosuppressive effects through multiple synergistic pathways, with specific mechanisms as follows: ①CTLA-4 expressed on the surface of Treg can bind to co-stimulatory molecules CD80/CD86 on antigen-presenting cells (APCs), competitively inhibiting CD28-mediated co-stimulatory signaling in T cells, while inducing APCs to express indoleamine 2,3-dioxygenase (IDO), thereby inhibiting T cell proliferation via metabolic pathways. ②Treg highly express IL-2R, which enables them to efficiently sequester IL-2 in the microenvironment, reducing the availability of this key cytokine required for conventional T cell (Tcon) activation, and thereby inhibiting the proliferation and functional exertion of Tcon. ③Treg can secrete multiple anti-inflammatory cytokines, such as IL-10, IL-35, and TGF-β, which suppress immune responses. ④CD39 and CD73 expressed on the surface of Treg can hydrolyze ATP in the microenvironment to generate adenosine; upon binding to the A2A adenosine receptor (A2AR) on Tcon, adenosine inhibit the activity of Tcon. ⑤Treg can induce apoptosis of Tcon by secreting granzyme and perforin.
Fig 2 Multilayered immunosuppressive mechanisms of Treg
| [1] | Ribatti D, Crivellato E. From horror autotoxicus to autoimmunity. An historical note[M]//Rezaei N. Translational autoimmunity: challenges for autoimmune diseases. London: Elsevier, 2023: 9-16. |
| [2] | Billingham R E, Brent L, Medawar P B. 'Actively acquired tolerance' of foreign cells [J]. Nature, 1953, 172(4379): 603-606. |
| [3] | Burnet F M. The immunological significance of the thymus: an extension of the clonal selection theory of immunity[J]. Australas Ann Med, 1962, 11: 79-91. |
| [4] | Gershon R K, Kondo K. Cell interactions in the induction of tolerance: the role of thymic lymphocytes[J]. Immunology, 1970, 18(5): 723-737. |
| [5] | Keating P, Cambrosio A. Helpers and suppressors: on fictional characters in immunology[J]. J Hist Biol, 1997, 30(3): 381-396. |
| [6] | Sakaguchi S, Sakaguchi N, Asano M, et al. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases[J]. J Immunol, 1995, 155(3): 1151-1164. |
| [7] | Leonard W J, Depper J M, Uchiyama T, et al. A monoclonal antibody that appears to recognize the receptor for human T-cell growth factor; partial characterization of the receptor[J]. Nature, 1982, 300(5889): 267-269. |
| [8] | Brunkow M E, Jeffery E W, Hjerrild K A, et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse[J]. Nat Genet, 2001, 27(1): 68-73. |
| [9] | Bennett C L, Christie J, Ramsdell F, et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3[J]. Nat Genet, 2001, 27(1): 20-21. |
| [10] | Wildin R S, Ramsdell F, Peake J, et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy[J]. Nat Genet, 2001, 27(1): 18-20. |
| [11] | Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3[J]. Science, 2003, 299(5609): 1057-1061. |
| [12] | Fontenot J D, Gavin M A, Rudensky A Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells[J]. Nat Immunol, 2003, 4(4): 330-336. |
| [13] | Khattri R, Cox T, Yasayko S A, et al. An essential role for Scurfin in CD4+CD25+ T regulatory cells[J]. Nat Immunol, 2003, 4(4): 337-342. |
| [14] | Lee H M, Bautista J L, Scott-Browne J, et al. A broad range of self-reactivity drives thymic regulatory T cell selection to limit responses to self[J]. Immunity, 2012, 37(3): 475-486. |
| [15] | Cozzo Picca C, Simons D M, Oh S, et al. CD4+CD25+Foxp3+ regulatory T cell formation requires more specific recognition of a self-peptide than thymocyte deletion[J]. Proc Natl Acad Sci U S A, 2011, 108(36): 14890-14895. |
| [16] | Owen D L, Mahmud S A, Sjaastad L E, et al. Thymic regulatory T cells arise via two distinct developmental programs[J]. Nat Immunol, 2019, 20(2): 195-205. |
| [17] | Kawakami R, Kitagawa Y, Chen K Y, et al. Distinct Foxp3 enhancer elements coordinate development, maintenance, and function of regulatory T cells[J]. Immunity, 2021, 54(5): 947-961.e8. |
| [18] | Brown C C, Rudensky A Y. Spatiotemporal regulation of peripheral T cell tolerance[J]. Science, 2023, 380(6644): 472-478. |
| [19] | Hill J A, Feuerer M, Tash K, et al. Foxp3 transcription-factor-dependent and -independent regulation of the regulatory T cell transcriptional signature[J]. Immunity, 2007, 27(5): 786-800. |
| [20] | Mukhopadhya I, Louis P. Gut microbiota-derived short-chain fatty acids and their role in human health and disease[J]. Nat Rev Microbiol, 2025, 23(10): 635-651. |
| [21] | Chen R Y, Zhang X M, Li B, et al. Progranulin-dependent repair function of regulatory T cells drives bone-fracture healing[J]. J Clin Invest, 2024, 135(2): e180679. |
| [22] | Chen W J, Jin W W, Hardegen N, et al. Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3[J]. J Exp Med, 2003, 198(12): 1875-1886. |
| [23] | Zheng S G, Gray J D, Ohtsuka K, et al. Generation ex vivo of TGF-β- producing regulatory T cells from CD4+CD25- precursors[J]. J Immunol, 2002, 169(8): 4183-4189. |
| [24] | Walker M R, Kasprowicz D J, Gersuk V H, et al. Induction of FoxP3 and acquisition of T regulatory activity by stimulated human CD4+CD25- T cells[J]. J Clin Invest, 2003, 112(9): 1437-1443. |
| [25] | Gavin M A, Torgerson T R, Houston E, et al. Single-cell analysis of normal and FOXP3-mutant human T cells: FOXP3 expression without regulatory T cell development[J]. Proc Natl Acad Sci U S A, 2006, 103(17): 6659-6664. |
| [26] | Chen K Y, Kibayashi T, Giguelay A, et al. Genome-wide CRISPR screen in human T cells reveals regulators of FOXP3[J]. Nature, 2025, 642(8066): 191-200. |
| [27] | Liu Q, Zhu F M, Liu X N, et al. Non-oxidative pentose phosphate pathway controls regulatory T cell function by integrating metabolism and epigenetics[J]. Nat Metab, 2022, 4(5): 559-574. |
| [28] | 童雪梅, 李斌. 从“免疫平衡”到“免疫震荡”: 2025年诺贝尔生理学或医学奖解读及展望[J]. 中国科学基金, 2025, 39(5): 791-793. |
| Tong X M, Li B. From "immune homeostasis" to "immune perturbation": insights and perspectives on the 2025 Nobel prize in physiology or medicine[J]. Bulletin of National Natural Science Foundation of China, 2025, 39(5): 791-793. | |
| [29] | Burchill M A, Yang J Y, Vogtenhuber C, et al. IL-2 receptor β-dependent STAT5 activation is required for the development of Foxp3+ regulatory T cells[J]. J Immunol, 2007, 178(1): 280-290. |
| [30] | Wu Y Q, Borde M, Heissmeyer V, et al. FOXP3 controls regulatory T cell function through cooperation with NFAT[J]. Cell, 2006, 126(2): 375-387. |
| [31] | Zheng Y, Josefowicz S, Chaudhry A, et al. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate[J]. Nature, 2010, 463(7282): 808-812. |
| [32] | Liu X N, Zhang W Q, Han Y C, et al. FOXP3+ regulatory T cell perturbation mediated by the IFNγ-STAT1-IFITM3 feedback loop is essential for anti-tumor immunity[J]. Nat Commun, 2024, 15(1): 122. |
| [33] | van Loosdregt J, Vercoulen Y, Guichelaar T, et al. Regulation of Treg functionality by acetylation-mediated Foxp3 protein stabilization[J]. Blood, 2010, 115(5): 965-974. |
| [34] | Yang J, Wei P, Barbi J, et al. The deubiquitinase USP44 promotes Treg function during inflammation by preventing FOXP3 degradation[J]. EMBO Rep, 2020, 21(9): e50308. |
| [35] | Wang L B, Cheng H, Wang X X, et al. Deubiquitination of aryl hydrocarbon receptor by USP21 negatively regulates T helper 17 cell differentiation[J]. J Leukoc Biol, 2024, 117(1): qiae148. |
| [36] | Li Z Y, Lin F, Zhuo C H, et al. PIM1 kinase phosphorylates the human transcription factor FOXP3 at serine 422 to negatively regulate its activity under inflammation[J]. J Biol Chem, 2014, 289(39): 26872-26881. |
| [37] | Cheng H, Wang L B, Yang B L, et al. Cutting edge: inhibition of glycogen synthase kinase 3 activity induces the generation and enhanced suppressive function of human IL-10+ FOXP3+-induced regulatory T cells[J]. J Immunol, 2020, 205(6): 1497-1502. |
| [38] | Huang Q R, Tian N, Zhang J F, et al. Nonclassical action of Ku70 promotes Treg-suppressive function through a FOXP3-dependent mechanism in lung adenocarcinoma[J]. J Clin Invest, 2025, 135(4): e191305. |
| [39] | Li D, Kong C, Tsun A, et al. miR-125a-5p decreases the sensitivity of Treg cells toward IL-6-mediated conversion by inhibiting IL-6R and STAT3 expression[J]. Sci Rep, 2015, 5: 14615. |
| [40] | Read S, Malmström V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation[J]. J Exp Med, 2000, 192(2): 295-302. |
| [41] | Takahashi T, Tagami T, Yamazaki S, et al. Immunologic self-tolerance maintained by CD25+CD4+ regulatory T cells constitutively expressing cytotoxic T lymphocyte-associated antigen 4[J]. J Exp Med, 2000, 192(2): 303-310. |
| [42] | Plitas G, Konopacki C, Wu K M, et al. Regulatory T cells exhibit distinct features in human breast cancer[J]. Immunity, 2016, 45(5): 1122-1134. |
| [43] | Jiang Z Y, Wang H Z, Wang X X, et al. TMED4 facilitates regulatory T cell suppressive function via ROS homeostasis in tumor and autoimmune mouse models[J]. J Clin Invest, 2024, 135(1): e179874. |
| [44] | Goldmann O, Nwofor O V, Chen Q, et al. Mechanisms underlying immunosuppression by regulatory cells[J]. Front Immunol, 2024, 15: 1328193. |
| [45] | Deng B L, Yang B L, Chen J Q, et al. Gallic acid induces T-helper-1-like Treg cells and strengthens immune checkpoint blockade efficacy[J]. J Immunother Cancer, 2022, 10(7): e004037. |
| [46] | Wang X X, Cheng H, Shen Y G, et al. Metabolic choice tunes Foxp3+ regulatory T cell function[J]. Adv Exp Med Biol, 2021, 1278: 81-94. |
| [47] | Xu N N, Zhu Y M, Han Y C, et al. Targeting MondoA-TXNIP restores antitumour immunity in lactic-acid-induced immunosuppressive microenvironment[J]. Nat Metab, 2025, 7(9): 1889-1904. |
| [48] | Grossman W J, Verbsky J W, Tollefsen B L, et al. Differential expression of granzymes A and B in human cytotoxic lymphocyte subsets and T regulatory cells[J]. Blood, 2004, 104(9): 2840-2848. |
| [49] | Nie P P, Cao Z F, Yu R X, et al. Targeting p97-Npl4 interaction inhibits tumor Treg cell development to enhance tumor immunity[J]. Nat Immunol, 2024, 25(9): 1623-1636. |
| [50] | Hu W, Wang Z M, Feng Y Q, et al. Regulatory T cells function in established systemic inflammation and reverse fatal autoimmunity[J]. Nat Immunol, 2021, 22(9): 1163-1174. |
| [51] | Jäger C, Dimitrova P, Sun Q, et al. Inducible protein degradation reveals inflammation-dependent function of the Treg cell lineage-defining transcription factor Foxp3[J]. Sci Immunol, 2025, 10(108): eadr7057. |
| [52] | Nie H, Zheng Y X, Li R S, et al. Phosphorylation of FOXP3 controls regulatory T cell function and is inhibited by TNF-α in rheumatoid arthritis[J]. Nat Med, 2013, 19(3): 322-328. |
| [53] | Chen Z J, Barbi J, Bu S R, et al. The ubiquitin ligase Stub1 negatively modulates regulatory T cell suppressive activity by promoting degradation of the transcription factor Foxp3[J]. Immunity, 2013, 39(2): 272-285. |
| [54] | Samanta A, Li B, Song X M, et al. TGF-β and IL-6 signals modulate chromatin binding and promoter occupancy by acetylated FOXP3[J]. Proc Natl Acad Sci U S A, 2008, 105(37): 14023-14027. |
| [55] | Guo Y X, Chen J N, Huang Y, et al. Increased infiltration of CD4+IL-17A+FOXP3+ T cells in Helicobacter pylori-induced gastritis[J]. Eur J Immunol, 2024, 54(3): e2350662. |
| [56] | Tian N, Liu Q, Li Y K, et al. Transketolase deficiency in adipose tissues protects mice from diet-induced obesity by promoting lipolysis[J]. Diabetes, 2020, 69(7): 1355-1367. |
| [57] | Ma X, Deng G S, Tian N, et al. Calycosin enhances Treg differentiation for alleviating skin inflammation in atopic dermatitis[J]. J Ethnopharmacol, 2024, 326: 117883. |
| [58] | Kidani Y, Nogami W, Yasumizu Y, et al. CCR8-targeted specific depletion of clonally expanded Treg cells in tumor tissues evokes potent tumor immunity with long-lasting memory[J]. Proc Natl Acad Sci U S A, 2022, 119(7): e2114282119. |
| [59] | Wen Y J, Xia Y, Yang X P, et al. CCR8: a promising therapeutic target against tumor-infiltrating regulatory T cells[J]. Trends Immunol, 2025, 46(2): 153-165. |
| [60] | Guo Y X, Xie F, Liu X, et al. Blockade of TNF-α/TNFR2 signalling suppresses colorectal cancer and enhances the efficacy of anti-PD1 immunotherapy by decreasing CCR8+ T regulatory cells[J]. J Mol Cell Biol, 2024, 16(6): mjad067. |
| [61] | Roider H G, Hoff S, Tseng S Y, et al. Selective depletion of tumor-infiltrating regulatory T cells with BAY 3375968, a novel Fc-optimized anti-CCR8 antibody[J]. Clin Exp Med, 2024, 24(1): 122. |
| [62] | Sakaguchi S, Yamaguchi T, Nomura T, et al. Regulatory T cells and immune tolerance[J]. Cell, 2008, 133(5): 775-787. |
| [63] | Wardell C M, Fung V C W, Chen E, et al. CAR Treg cells mediate linked suppression and infectious tolerance in islet transplantation in mice[J]. Sci Transl Med, 2025, 17(812): eadp6519. |
| [64] | Tahvildari M, Dana R. Low-dose IL-2 therapy in transplantation, autoimmunity, and inflammatory diseases[J]. J Immunol, 2019, 203(11): 2749-2755. |
| [65] | Gu W, Ge R R, Zhu F M, et al. PARP-1 inhibitor——AG14361 suppresses acute allograft rejection via stabilizing CD4+FoxP3+ regulatory T cells[J]. Pathol Res Pract, 2020, 216(8): 153021. |
| [66] | Li Y Y, Lu Y, Lin S H, et al. Insulin signaling establishes a developmental trajectory of adipose regulatory T cells[J]. Nat Immunol, 2021, 22(9): 1175-1185. |
| [67] | Tong L F, Chen Z B, Li Y Y, et al. Transketolase promotes MAFLD by limiting inosine-induced mitochondrial activity[J]. Cell Metab, 2024, 36(5): 1013-1029.e5. |
| [68] | González H, Pacheco R. T-cell-mediated regulation of neuroinflammation involved in neurodegenerative diseases[J]. J Neuroinflammation, 2014, 11: 201. |
| [69] | Li M, Kui X Y, Yang S J, et al. LdIL-2 treatment in ASD: a novel immunotherapeutic approach targeting Th/Treg dysfunction and neuroinflammation[J]. Transl Psychiatry, 2025, 15(1): 376. |
| [70] | Laukova M, Glatman Zaretsky A. Regulatory T cells as a therapeutic approach for inflammatory bowel disease[J]. Eur J Immunol, 2023, 53(2): e2250007. |
| [71] | Brighenti S, Joosten S A. Friends and foes of tuberculosis: modulation of protective immunity[J]. J Intern Med, 2018: 10.1111/joim.12778. |
| [72] | Maizels R M, Smith K A. Regulatory T cells in infection[J]. Adv Immunol, 2011, 112: 73-136. |
| [73] | Li X Y, Fu S C, Cheng H, et al. Differentiation of type 17 mucosal-associated invariant T cells in circulation contributes to the severity of sepsis[J]. Am J Pathol, 2024, 194(7): 1248-1261. |
| [74] | Bender C, Wiedeman A E, Hu A, et al. A phase 2 randomized trial with autologous polyclonal expanded regulatory T cells in children with new-onset type 1 diabetes[J]. Sci Transl Med, 2024, 16(746): eadn2404. |
| [75] | Silverberg J I, Rosmarin D, Chovatiya R, et al. The regulatory T cell-selective interleukin-2 receptor agonist rezpegaldesleukin in the treatment of inflammatory skin diseases: two randomized, double-blind, placebo-controlled phase 1b trials[J]. Nat Commun, 2024, 15(1): 9230. |
| [76] | Porret R, Alcaraz-Serna A, Peter B, et al. T cell receptor precision editing of regulatory T cells for celiac disease[J]. Sci Transl Med, 2025, 17(790): eadr8941. |
| [77] | Tian N, Cheng H, Du Y, et al. Cannabinoid receptor 2 selective agonist alleviates systemic sclerosis by inhibiting Th2 differentiation through JAK/SOCS3 signaling[J]. J Autoimmun, 2024, 147: 103233. |
| [78] | Jiang Z Y, Huang Q R, Chang Y J, et al. LILRB2 promotes immune escape in breast cancer cells via enhanced HLA-A degradation[J]. Cell Oncol (Dordr), 2024, 47(5): 1679-1696. |
| [1] | 胡飞, 蔡晓涵, 程睿, 季诗雨, 苗嘉欣, 朱晏, 范广建. 骨肉瘤免疫微环境及其免疫治疗临床转化研究进展[J]. 上海交通大学学报(医学版), 2024, 44(7): 814-821. |
| [2] | 张烨晟, 杨易静, 黄依雯, 施珑玙, 王曼媛, 陈思思. 肿瘤微环境免疫细胞调节肿瘤细胞耐药性的研究进展[J]. 上海交通大学学报(医学版), 2024, 44(7): 830-838. |
| [3] | 韩婷, 吕纯鑫, 卓萌, 夏青, 刘腾飞, 吴秀奇, 林晓琳, 肖秀英. 进展期胃癌免疫治疗不良反应的相关因素及预后分析[J]. 上海交通大学学报(医学版), 2022, 42(8): 1053-1061. |
| [4] | 阿婷曦, 邵春益, 傅瑶. 调节性T细胞在眼表疾病中作用的研究进展[J]. 上海交通大学学报(医学版), 2022, 42(8): 1145-1150. |
| [5] | 柏文心, 赵爱民. 不明原因复发性流产的免疫发病机制研究进展[J]. 上海交通大学学报(医学版), 2021, 41(10): 1371-1377. |
| [6] | 袁瑞雪, 傅迎美, 禹顺英. 辅助性T细胞17和调节性T细胞在抑郁症中的作用机制研究进展[J]. 上海交通大学学报(医学版), 2021, 41(10): 1384-1388. |
| [7] | 袁 芸,张洪铭,黄 慧. 牙周炎对大鼠外周血中辅助性T细胞分群的影响[J]. 上海交通大学学报(医学版), 2020, 40(10): 1360-1364. |
| [8] | 熊苗,徐亮,李莉,刘阳,周芳芳,王军,朱洁萍 . 新型免疫抑制剂 FTY720 通过阻断 S1P 信号通路诱导母胎免疫 耐受的实验研究[J]. 上海交通大学学报(医学版), 2017, 37(10): 1332-. |
| [9] | 熊苗,朱洁萍,李莉,刘阳,季玉琴,王军. 过继转移FTY720-DC对自然流产模型孕鼠胚胎丢失率的影响[J]. 上海交通大学学报(医学版), 2016, 36(6): 793-. |
| [10] | 张晓欣,赵爱民. CCL20对CBA/J小鼠CCR6+调节性T细胞及妊娠结局的影响[J]. 上海交通大学学报(医学版), 2016, 36(2): 147-. |
| [11] | 李维宏,牟晓玲. 脂联素对不明原因复发性流产患者外周血Th17/Treg细胞分群及细胞因子水平的影响[J]. 上海交通大学学报(医学版), 2015, 35(7): 1004-. |
| [12] | 孙蔚,朱海超,甘建和,等. 戊型病毒性肝炎患者外周血Th17、Treg、Th17/Treg变化及其临床意义[J]. 上海交通大学学报(医学版), 2015, 35(11): 1647-. |
| [13] | 邱 添,滕银成,徐 亮. 过继性转移TGF-β诱导CD4+CD25+调节性T细胞对自然流产模型小鼠胚胎丢失率的影响[J]. 上海交通大学学报(医学版), 2014, 34(8): 1120-. |
| [14] | 陈春媛,牛 鑫,汪 泱. Exosomes与肾脏生理及疾病[J]. 上海交通大学学报(医学版), 2014, 34(4): 556-. |
| [15] | 柳龙根, 申红玉, 叶春艳, 等. 慢性丙型肝炎患者外周血CD4+CD25high调节性T细胞与抗病毒疗效的关系[J]. , 2012, 32(12): 1610-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||