
上海交通大学学报(医学版) ›› 2026, Vol. 46 ›› Issue (3): 368-376.doi: 10.3969/j.issn.1674-8115.2026.03.011
• 综述 • 上一篇
赵旻炅1, 陈铃芳1, 胡苗清1, 冯杰1,2, 聂宇1,2( )
)
收稿日期:2025-07-31
接受日期:2025-12-01
出版日期:2026-03-28
发布日期:2026-03-30
通讯作者:
聂 宇,研究员,博士;电子信箱:nieyuniverse@126.com。基金资助:
Zhao Minjiong1, Chen Lingfang1, Hu Miaoqing1, Feng Jie1,2, Nie Yu1,2( )
)
Received:2025-07-31
Accepted:2025-12-01
Online:2026-03-28
Published:2026-03-30
Contact:
Nie Yu, E-mail: nieyuniverse@126.com.Supported by:摘要:
缺血性心脏病特别是心肌梗死可导致大量心肌细胞不可逆性丧失。由于成年哺乳动物心肌细胞增殖能力高度受限,心肌受损区域通常被纤维化瘢痕组织取代,进而引发心室重构和心力衰竭,对健康造成严重威胁。近年来,诱导内源性心肌再生已成为改善心肌损伤预后的潜在治疗策略,而免疫调控机制在其中发挥核心作用。心肌损伤后,机体迅速启动复杂的炎症免疫级联反应。研究表明,炎症反应的时序与强度是决定组织修复结局的关键。在具有再生能力的模型中炎症反应启动迅速且消退及时,而成年个体则常出现炎症消退延迟与免疫失调,导致纤维化瘢痕形成。在此过程中,多种免疫细胞(如巨噬细胞、中性粒细胞、T细胞等)凭借其高度异质性特征、时序性浸润模式及微环境特异性分布,通过细胞间直接接触或旁分泌途径等方式,精细调控心肌细胞增殖与组织修复进程。这些免疫调控机制相互协调,形成一个动态促进心肌再生的交互网络。该综述系统阐述了各类免疫细胞在心肌损伤后再生修复过程中的应答特征及其调控机制,以期为缺血性心脏病的临床治疗提供新的思路与策略。
中图分类号:
赵旻炅, 陈铃芳, 胡苗清, 冯杰, 聂宇. 免疫细胞调控心肌缺血损伤后再生的研究进展[J]. 上海交通大学学报(医学版), 2026, 46(3): 368-376.
Zhao Minjiong, Chen Lingfang, Hu Miaoqing, Feng Jie, Nie Yu. Research progress on immune cells regulation of cardiac regeneration after ischemic myocardial injury[J]. Journal of Shanghai Jiao Tong University (Medical Science), 2026, 46(3): 368-376.
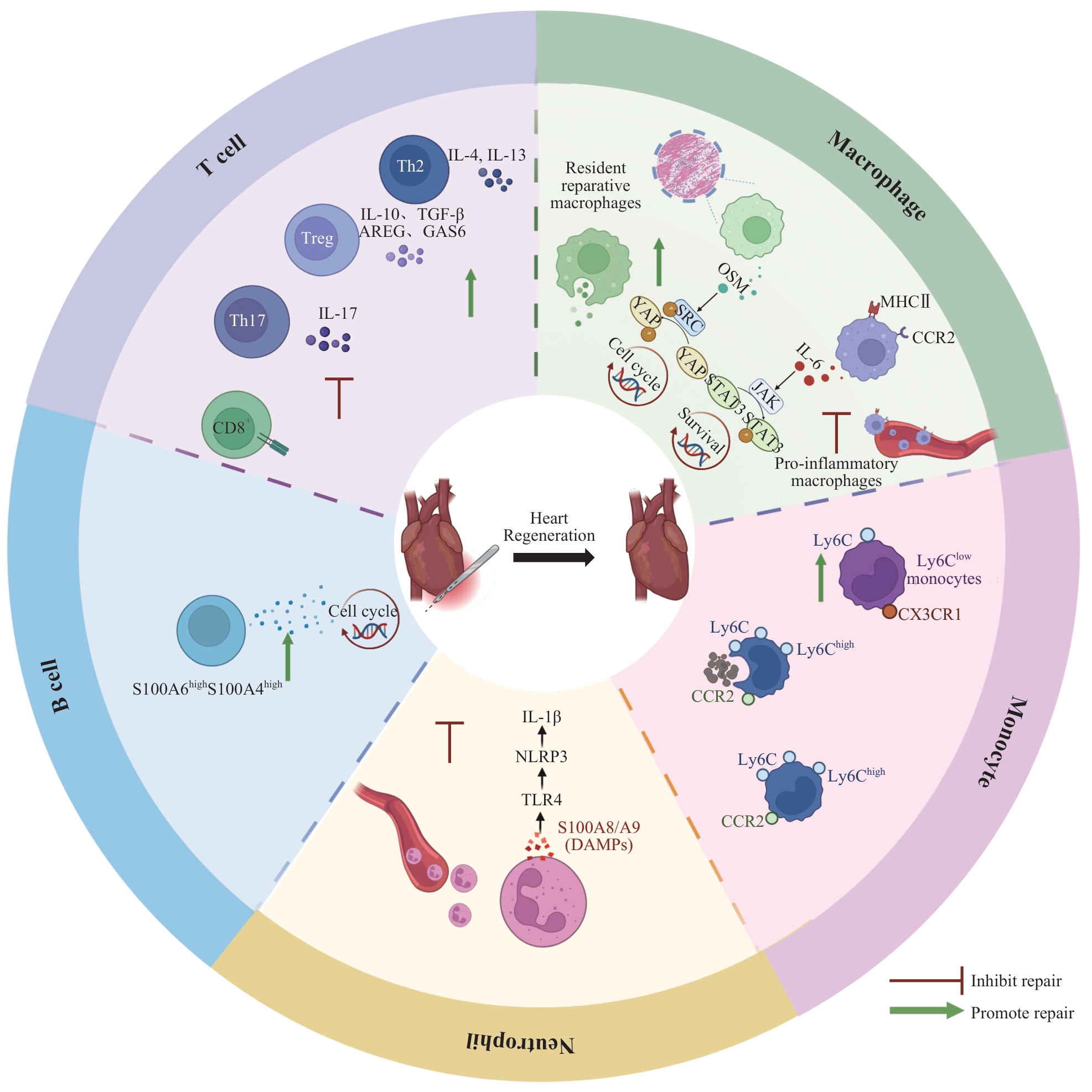
图1 参与心肌损伤后修复与再生的免疫细胞及其效应分子Note: The outer ring sequentially illustrates neutrophils, monocytes (Ly6ChighCCR2+, Ly6Clow), macrophages (CCR2- reparative, CCR2+ pro-inflammatory), T cells (CD8+, Th1/Th17, Treg, Th2), and B cells. Neutrophils are rapidly recruited post-injury to clear necrotic debris and amplify acute inflammation via the S100A8/A9-TLR4-NLRP3-IL-1β axis, subsequently contributing to the resolution of inflammation. Early-stage Ly6ChighCCR2+ monocytes are primarily responsible for phagocytosis and clearance; subsequently, Ly6Clow monocytes and their differentiated reparative macrophages synergistically promote tissue reconstruction and angiogenesis. Resident CCR2- reparative macrophages directly drive cardiomyocyte dedifferentiation and proliferation via the OSM-OSMR/gp130-SRC-YAP (Y357) pathway, while also promoting homeostasis and angiogenesis through efferocytosis and IGF1/IGF2 secretion. Transiently appearing CCR2+ pro-inflammatory macrophages secrete IL-6, serving as a critical trigger signal for the regenerative response. In adaptive immunity, CD8+ T cells and Th1/Th17 subsets (associated with IFN-γ and IL-17) inhibit regeneration. Conversely, Tregs (secreting IL-10, TGF-β, AREG, and GAS6) and Th2 cells (secreting IL-4 and IL-13) support cardiomyocyte proliferation and functional recovery by remodelling the immune microenvironment and exerting direct or indirect regulatory effects. Neonatal B cells (including the S100A6highS100A4high subset) secrete various factors to promote cardiomyocyte proliferation.
Fig 1 Immune cells and their effector molecules involved in repair and regeneration after myocardial injury
| [1] | Zhang L, Tong Z, Han R Z, et al. Global, regional, and national burdens of ischemic heart disease attributable to smoking from 1990 to 2019[J]. J Am Heart Assoc, 2023, 12(3): e028193. |
| [2] | Byrne R A, Rossello X, Coughlan J J, et al. 2023 ESC guidelines for the management of acute coronary syndromes[J]. Eur Heart J, 2023, 44(38): 3720-3826. |
| [3] | Rao S V, O´Donoghue M L, Ruel M, et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI guideline for the management of patients with acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Joint Committee on clinical practice guidelines[J]. Circulation, 2025, 151(13): e771-e862. |
| [4] | Bergmann O, Zdunek S, Felker A, et al. Dynamics of cell generation and turnover in the human heart[J]. Cell, 2015, 161(7): 1566-1575. |
| [5] | Poss K D, Wilson L G, Keating M T. Heart regeneration in zebrafish[J]. Science, 2002, 298(5601): 2188-2190. |
| [6] | Porrello E R, Mahmoud A I, Simpson E, et al. Transient regenerative potential of the neonatal mouse heart[J]. Science, 2011, 331(6020): 1078-1080. |
| [7] | Derks W, Rode J, Collin S, et al. A latent cardiomyocyte regeneration potential in human heart disease[J]. Circulation, 2025, 151(3): 245-256. |
| [8] | Hilgendorf I, Frantz S, Frangogiannis N G. Repair of the infarcted heart: cellular effectors, molecular mechanisms and therapeutic opportunities[J]. Circ Res, 2024, 134(12): 1718-1751. |
| [9] | Cortada E, Yao J, Xia Y, et al. Cross-species single-cell RNA-seq analysis reveals disparate and conserved cardiac and extracardiac inflammatory responses upon heart injury[J]. Commun Biol, 2024, 7(1): 1611. |
| [10] | Han C Y, Nie Y, Lian H, et al. Acute inflammation stimulates a regenerative response in the neonatal mouse heart[J]. Cell Res, 2015, 25(10): 1137-1151. |
| [11] | Dick S A, Wong A, Hamidzada H, et al. Three tissue resident macrophage subsets coexist across organs with conserved origins and life cycles[J]. Sci Immunol, 2022, 7(67): eabf7777. |
| [12] | Weinberger T, Denise M, Joppich M, et al. Resident and recruited macrophages differentially contribute to cardiac healing after myocardial ischemia[J]. eLife, 2024, 12: RP89377. |
| [13] | Chen R K, Zhang H R, Tang B T, et al. Macrophages in cardiovascular diseases: molecular mechanisms and therapeutic targets[J]. Signal Transduct Target Ther, 2024, 9(1): 130. |
| [14] | Bajpai G, Schneider C, Wong N, et al. The human heart contains distinct macrophage subsets with divergent origins and functions[J]. Nat Med, 2018, 24(8): 1234-1245. |
| [15] | Li Y D, Feng J, Song S, et al. gp130 controls cardiomyocyte proliferation and heart regeneration[J]. Circulation, 2020, 142(10): 967-982. |
| [16] | Fu M Z, Jia S T, Xu L H, et al. Single-cell multiomic analysis identifies macrophage subpopulations in promoting cardiac repair[J]. J Clin Invest, 2024, 134(19): e175297. |
| [17] | Jung M, Ma Y G, Iyer R P, et al. IL-10 improves cardiac remodeling after myocardial infarction by stimulating M2 macrophage polarization and fibroblast activation[J]. Basic Res Cardiol, 2017, 112(3): 33. |
| [18] | Isidoro C A, Deniset J F. The role of macrophage subsets in and around the heart in modulating cardiac homeostasis and pathophysiology[J]. Front Immunol, 2023, 14: 1111819. |
| [19] | Zhou Y, Stevis P E, Cao J, et al. Structures of complete extracellular assemblies of type I and type Ⅱ oncostatin M receptor complexes[J]. Nat Commun, 2024, 15(1): 9776. |
| [20] | Liu S J, Li R G, Martin J F. The cell-autonomous and non-cell-autonomous roles of the Hippo pathway in heart regeneration[J]. J Mol Cell Cardiol, 2022, 168: 98-106. |
| [21] | Xiao Y, Zhang H, Liu X, et al. Medium from human iPSC-derived primitive macrophages promotes adult cardiomyocyte proliferation and cardiac regeneration[J]. Nat Commun, 2025, 16(1): 3012. |
| [22] | Jiang Y L, Niu S W, Lin Z, et al. Injectable hydrogel with dual-sensitive behavior for targeted delivery of oncostatin M to improve cardiac restoration after myocardial infarction[J]. J Mater Chem B, 2022, 10(34): 6514-6531. |
| [23] | Li Y D, Li H T, Pei J Q, et al. Transplantation of murine neonatal cardiac macrophage improves adult cardiac repair[J]. Cell Mol Immunol, 2021, 18(2): 492-494. |
| [24] | Sun K, Li Y Y, Jin J. A double-edged sword of immuno-microenvironment in cardiac homeostasis and injury repair[J]. Signal Transduct Target Ther, 2021, 6(1): 79. |
| [25] | Miyake K, Ito J, Takahashi K, et al. Single-cell transcriptomics identifies the differentiation trajectory from inflammatory monocytes to pro-resolving macrophages in a mouse skin allergy model[J]. Nat Commun, 2024, 15(1): 1666. |
| [26] | Trzebanski S, Kim J S, Larossi N, et al. Classical monocyte ontogeny dictates their functions and fates as tissue macrophages[J]. Immunity, 2024, 57(6): 1225-1242.e6. |
| [27] | Gallerand A, Dolfi B, Stunault M I, et al. Glucose metabolism controls monocyte homeostasis and migration but has no impact on atherosclerosis development in mice[J]. Nat Commun, 2024, 15(1): 9027. |
| [28] | Thierry G R, Baudon E M, Bijnen M, et al. Non-classical monocytes scavenge the growth factor CSF1 from endothelial cells in the peripheral vascular tree to ensure survival and homeostasis[J]. Immunity, 2024, 57(9): 2108-2121. |
| [29] | Minton K. Non-classical monocytes have the support of the whole vascular tree[J]. Nat Rev Immunol, 2024, 24(9): 619. |
| [30] | Nahrendorf M, Swirski F K, Aikawa E, et al. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions[J]. J Exp Med, 2007, 204(12): 3037-3047. |
| [31] | Chen Z W, Cai D C, Xie Y F, et al. Triiodothyronine induces a proinflammatory monocyte/macrophage profile and impedes cardiac regeneration[J]. J Mol Cell Cardiol, 2024, 191: 7-11. |
| [32] | He S, Chousterman B G, Fenn A, et al. Lp-PLA2 antagonizes left ventricular healing after myocardial infarction by impairing the appearance of reparative macrophages[J]. Circ Heart Fail, 2015, 8(5): 980-987. |
| [33] | Rizzo G, Gropper J, Piollet M, et al. Dynamics of monocyte-derived macrophage diversity in experimental myocardial infarction[J]. Cardiovasc Res, 2023, 119(3): 772-785. |
| [34] | Kubota A, Frangogiannis N G. Macrophages in myocardial infarction[J]. Am J Physiol Cell Physiol, 2022, 323(4): C1304-C1324. |
| [35] | Kim C, Kim H, Sim W S, et al. Spatiotemporal control of neutrophil fate to tune inflammation and repair for myocardial infarction therapy[J]. Nat Commun, 2024, 15(1): 8481. |
| [36] | Cohen C D, Rousseau S T, Bermea K C, et al. Myocardial immune cells: the basis of cardiac immunology[J]. J Immunol, 2023, 210(9): 1198-1207. |
| [37] | Kologrivova I, Shtatolkina M, Suslova T, et al. Cells of the immune system in cardiac remodeling: main players in resolution of inflammation and repair after myocardial infarction[J]. Front Immunol, 2021, 12: 664457. |
| [38] | Sreejit G, Abdel-Latif A, Athmanathan B, et al. Neutrophil-derived S100A8/A9 amplify granulopoiesis after myocardial infarction[J]. Circulation, 2020, 141(13): 1080-1094. |
| [39] | Zhou X H, Zhang C Y, Wu X Y, et al. Dusp6 deficiency attenuates neutrophil-mediated cardiac damage in the acute inflammatory phase of myocardial infarction[J]. Nat Commun, 2022, 13(1): 6672. |
| [40] | Wang X K, Xu Y, Yu C Q, et al. Periodontitis-related myocardial fibrosis by expansion of collagen-producing SiglecF+ neutrophils[J]. Eur Heart J, 2025, 46(23): 2223-2238. |
| [41] | He L, Liu R Q, Yue H H, et al. Interaction between neutrophil extracellular traps and cardiomyocytes contributes to atrial fibrillation progression[J]. Signal Transduct Target Ther, 2023, 8(1): 279. |
| [42] | Zhang N, Aiyasiding X, Li W J, et al. Neutrophil degranulation and myocardial infarction[J]. Cell Commun Signal, 2022, 20(1): 50. |
| [43] | Petzold T, Zhang Z, Ballesteros I, et al. Neutrophil “plucking” on megakaryocytes drives platelet production and boosts cardiovascular disease[J]. Immunity, 2022, 55(12): 2285-2299. |
| [44] | Thakur M, Junho C V C, Bernhard S M, et al. NETs-induced thrombosis impacts on cardiovascular and chronic kidney disease[J]. Circ Res, 2023, 132(8): 933-949. |
| [45] | Mangold A, Ondracek A S, Hofbauer T M, et al. Culprit site extracellular DNA and microvascular obstruction in ST-elevation myocardial infarction[J]. Cardiovasc Res, 2022, 118(8): 2006-2017. |
| [46] | Chalise U, Becirovic-Agic M, Lindsey M L. Neutrophil crosstalk during cardiac wound healing after myocardial infarction[J]. Curr Opin Physiol, 2021, 24: 100485. |
| [47] | Santos-Zas I, Lemarié J, Zlatanova I, et al. Cytotoxic CD8+ T cells promote granzyme B-dependent adverse post-ischemic cardiac remodeling[J]. Nat Commun, 2021, 12(1): 1483. |
| [48] | Alcaide P, Kallikourdis M, Emig R, et al. Myocardial inflammation in heart failure with reduced and preserved ejection fraction[J]. Circ Res, 2024, 134(12): 1752-1766. |
| [49] | Wang D Z, Weng X Y, Yue W H, et al. CD8 T cells promote heart failure progression in mice with preexisting left ventricular dysfunction[J]. Front Immunol, 2024, 15: 1472133. |
| [50] | Alshoubaki Y K, Nayer B, Lu Y Z, et al. Tregs delivered post-myocardial infarction adopt an injury-specific phenotype promoting cardiac repair via macrophages in mice[J]. Nat Commun, 2024, 15(1): 6480. |
| [51] | Sun L N, Su Y H, Jiao A J, et al. T cells in health and disease[J]. Signal Transduct Target Ther, 2023, 8(1): 235. |
| [52] | Li J T, Liang C, Yang K Y, et al. Specific ablation of CD4+ T-cells promotes heart regeneration in juvenile mice[J]. Theranostics, 2020, 10(18): 8018-8035. |
| [53] | Dolejsi T, Delgobo M, Schuetz T, et al. Adult T-cells impair neonatal cardiac regeneration[J]. Eur Heart J, 2022, 43(28): 2698-2709. |
| [54] | Bakhshian Nik A, Alvarez-Argote S, O′Meara C C. Interleukin 4/13 signaling in cardiac regeneration and repair[J]. Am J Physiol Heart Circ Physiol, 2022, 323(5): H833-H844. |
| [55] | Molofsky A B, Locksley R M. The ins and outs of innate and adaptive type 2 immunity[J]. Immunity, 2023, 56(4): 704-722. |
| [56] | Paddock S J, Swift S K, Alencar-Almeida V, et al. IL4Rα signaling promotes neonatal cardiac regeneration and cardiomyocyte cell cycle activity[J]. J Mol Cell Cardiol, 2021, 161: 62-74. |
| [57] | Alvarez-Argote S, Paddock S J, Flinn M A, et al. IL-13 promotes functional recovery after myocardial infarction via direct signaling to macrophages[J]. JCI Insight, 2024, 9(2): e172702. |
| [58] | Liu J, Liu F L, Liang T T, et al. The roles of Th cells in myocardial infarction[J]. Cell Death Discov, 2024, 10(1): 287. |
| [59] | Huang L Q. The role of IL-17 family cytokines in cardiac fibrosis[J]. Front Cardiovasc Med, 2024, 11: 1470362. |
| [60] | Takahashi J, Yamamoto M, Yasukawa H, et al. Interleukin-22 directly activates myocardial STAT3 (signal transducer and activator of transcription-3) signaling pathway and prevents myocardial ischemia reperfusion injury[J]. J Am Heart Assoc, 2020, 9(8): e014814. |
| [61] | Yamamoto M, Yasukawa H, Takahashi J, et al. Endogenous interleukin-22 prevents cardiac rupture after myocardial infarction in mice[J]. PLoS One, 2023, 18(6): e0286907. |
| [62] | Liu F, Chen Y Q, Qin D M, et al. Interleukin-22 inhibits cardiac fibrosis by regulating fibroblast metabolic reprogramming in myocardial infarction[J]. Pathol Res Pract, 2024, 256: 155256. |
| [63] | Yang Y, Li X Y, Li L C, et al. γδ T/interleukin-17A contributes to the effect of maresin conjugates in tissue regeneration 1 on lipopolysaccharide-induced cardiac injury[J]. Front Immunol, 2021, 12: 674542. |
| [64] | Wang P, Kljavin N, Nguyen T T T, et al. Adrenergic nerves regulate intestinal regeneration through IL-22 signaling from type 3 innate lymphoid cells[J]. Cell Stem Cell, 2023, 30(9): 1166-1178. |
| [65] | Weirather J, Hofmann U D, Beyersdorf N, et al. Foxp3+ CD4+ T cells improve healing after myocardial infarction by modulating monocyte/macrophage differentiation[J]. Circ Res, 2014, 115(1): 55-67. |
| [66] | Nayer B, Tan J L, Alshoubaki Y K, et al. Local administration of regulatory T cells promotes tissue healing[J]. Nat Commun, 2024, 15(1): 7863. |
| [67] | Baccouche B M, Elde S, Wang H, et al. Structural, angiogenic, and immune responses influencing myocardial regeneration: a glimpse into the crucible[J]. NPJ Regen Med, 2024, 9(1): 18. |
| [68] | Wang Y, Li J, Zhang Y, et al. AREG+ regulatory T cells mediating myocardial repair and neovascularization after myocardial infarction[J]. Mol Med, 2025, 31(1): 229. |
| [69] | Courey-Ghaouzi A D, Kleberg L, Sundling C. Alternative B cell differentiation during infection and inflammation[J]. Front Immunol, 2022, 13: 908034. |
| [70] | Tan Y, Duan X W, Wang B, et al. Murine neonatal cardiac B cells promote cardiomyocyte proliferation and heart regeneration[J]. NPJ Regen Med, 2023, 8(1): 7. |
| [71] | Jiao J, He S J, Wang Y Q, et al. Regulatory B cells improve ventricular remodeling after myocardial infarction by modulating monocyte migration[J]. Basic Res Cardiol, 2021, 116(1): 46. |
| [72] | Mo F R, Luo Y, Yan Y L, et al. Are activated B cells involved in the process of myocardial fibrosis after acute myocardial infarction? An in vivo experiment[J]. BMC Cardiovasc Disord, 2021, 21(1): 5. |
| [73] | Marinković G, Koenis D S, de Camp L, et al. S100A9 links inflammation and repair in myocardial infarction[J]. Circ Res, 2020, 127(5): 664-676. |
| [74] | Sun Y Y, Pinto C, Camus S, et al. Splenic marginal zone B lymphocytes regulate cardiac remodeling after acute myocardial infarction in mice[J]. J Am Coll Cardiol, 2022, 79(7): 632-647. |
| [75] | de Winter N, Ji J H, Sintou A, et al. Persistent transcriptional changes in cardiac adaptive immune cells following myocardial infarction: new evidence from the re-analysis of publicly available single cell and nuclei RNA-sequencing data sets[J]. J Mol Cell Cardiol, 2024, 192: 48-64. |
| [76] | Yu X, Newland S A, Zhao T X, et al. Innate lymphoid cells promote recovery of ventricular function after myocardial infarction[J]. J Am Coll Cardiol, 2021, 78(11): 1127-1142. |
| [1] | 韩珍, 王浩, 苏秀秀, 方际. 金丝桃素通过抑制NF-κB信号通路减轻糖尿病肾病足细胞损伤[J]. 上海交通大学学报(医学版), 2026, 46(1): 43-53. |
| [2] | 杨全军, 柏丁源, 周雨萱, 白露, 郭澄. 异柠檬酸脱氢酶1突变介导D-2-羟基戊二酸代谢重编程在肿瘤免疫调控中的作用及相关药物研发进展[J]. 上海交通大学学报(医学版), 2025, 45(9): 1239-1248. |
| [3] | PANDIT Roshan, 卢君瑶, 何立珩, 包玉洁, 季萍, 陈颖盈, 许洁, 王颖. 肿瘤坏死因子-α在新型冠状病毒感染合并肾损伤中的作用[J]. 上海交通大学学报(医学版), 2025, 45(1): 1-10. |
| [4] | 衣文婧, 范宜璇, 仇佳泰, 付晓燕, 刘梅芳. β-抑制蛋白1在炎症反应调控中作用的研究进展[J]. 上海交通大学学报(医学版), 2024, 44(12): 1593-1600. |
| [5] | 宣臻全, 陈轩祎, 姚志荣. 神经免疫紊乱在特应性皮炎中的作用研究进展[J]. 上海交通大学学报(医学版), 2023, 43(8): 1049-1055. |
| [6] | 王青, 韩晓, 张晓波. 表观遗传修饰调控肺炎免疫应答的研究进展[J]. 上海交通大学学报(医学版), 2023, 43(7): 931-938. |
| [7] | 过丽强, 赵世天, 舒冰. Notch信号通路在骨折愈合过程中作用的研究进展[J]. 上海交通大学学报(医学版), 2023, 43(2): 222-229. |
| [8] | 林家俞, 秦洁洁, 蒋玲曦. 肿瘤微环境中免疫细胞的代谢研究进展[J]. 上海交通大学学报(医学版), 2022, 42(8): 1122-1130. |
| [9] | 王昊, 王然, 巴乾. 食品中二氧化钛纳米材料对消化道组织及肠道微生物群影响的研究进展[J]. 上海交通大学学报(医学版), 2022, 42(2): 225-229. |
| [10] | 高 涵,张 萍. 子宫内膜异位症的免疫学相关研究进展[J]. 上海交通大学学报(医学版), 2020, 40(11): 1544-1549. |
| [11] | 周 晗,杨晓笙,廖陈龙,张文川. 糖尿病足溃疡相关基因与免疫细胞特征分析[J]. 上海交通大学学报(医学版), 2020, 40(10): 1354-1359. |
| [12] | 王 齐1*,朱冠娅2*,谢 挺1,葛 奎1,牛轶雯3. ATP代谢及嘌呤信号受体在糖尿病创面愈合炎症反应阶段的变化[J]. 上海交通大学学报(医学版), 2020, 40(1): 10-. |
| [13] | 储维薇,徐洁颖,李尚,翟君钰,杜艳芝. 脱氢表雄酮诱导的多囊卵巢综合征模型大鼠的肠道菌群研究[J]. 上海交通大学学报(医学版), 2019, 39(9): 975-. |
| [14] | 陆海洋,赵维莅. 胃肠道微生物在肿瘤发生中的作用[J]. 上海交通大学学报(医学版), 2019, 39(9): 1083-. |
| [15] | 范霞 1,夏碧丽 2,吕霖 1,徐梦莎 3,李佳茵 1,何平 1. 小鼠巨噬细胞对钩端螺旋体 56606v和 56606a的吞噬及炎症应答的比较研究[J]. 上海交通大学学报(医学版), 2019, 39(1): 16-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||